Histiobalantium natans
Claparede & Lachmann, 1858
Most likely ID: n.a.
Synonym: n.a.
Sampling location: Simmelried
Phylogenetic tree: Histiobalantium natans
Diagnosis:
- body broadly ovate or ellipsoidal, dorsally hemispherical, ventrally flat
- length 55–100 µm
- on average seven contractile vacuoles, four in left side, three in right.
- usually 2 macronuclear nodules, irregularly shaped
- each macronuclear nodule with one micronucleus
- large oral apparatus
- extrusomes slightly curved rods with rounded ends, 3.5–4.5 µm long
- cytoplasm studded with greenish and brownish cleptoplasts, 3–6 µm in size
- somatic ciliation with several elongated tactile cilia, projecting radially in resting specimens
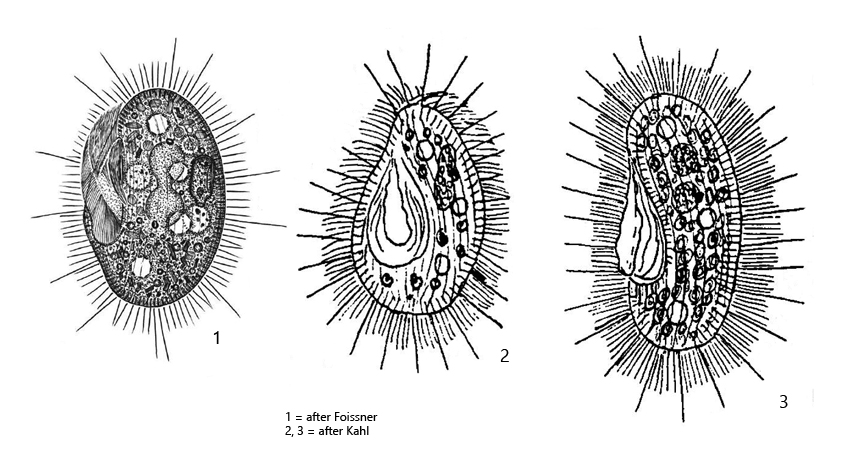
I find Histiobalantium natans regularly, but rarely in the Simmelried. In my other localities I have not found this species so far.
Kahl describes two forms of Histiobalantium natans. A somewhat stockier, green form Histiobalantium natans viridis (Kahl, 1931) and an elongated, bean-shaped form Histiobalantium natans nigricans (Kahl, 1931), which appears black due to numerous reserve grains in the cytoplasm. The green form Histiobalantium natans viridis was redescribed in 2009 by Foissner et al. and they described also the two forms “viridis” and “nigricans”. When the green form is maintained in culture, the chloroplasts are digested after one week. Nevertheless, these specimens do not assume an elongated, bean-like form, nor do they store any substantial amounts of reserve material. Thus, a transition between these forms has never been observed. Therefore, according to Foissner et al. it cannot be excluded that Histiobalantium natans nigricans is a distinct species. However, only further investigations can show whether this is the case.
In the Simmelried I find both forms and therefore I keep the designations of Kahl for the time being. I could never observe transitional forms between “viridis” and “nigricans”, so in my opinion they are truly two species.
The green form Histiobalantium natans viridis (s. drawings 1 and 2 above) is filled with chloroplasts. These are often yellowish or brown in color. As Foissner et al. noted, these chloroplasts have no cell wall of their own. Therefore, they must be cleptoplasts, i.e. chloroplasts extracted from prey organisms and transferred into the cytoplasm. Since brownish colored cleptoplasts are also often found in Histiobalantium natans viridis, they probably originate from dinoflagellates, while the green cleptoplasts originate from most leikely from euglenids. Esteban et al. (2008) performed experiments on cultures of Histiobalantium natans to clarify the origin of the cleptoplasts. They performed feeding experiments with different algae and euglenids and found that the chloroplasts of Phacus suecicus were transferred into the cytoplasm of Histiobalantium, while other algae (e.g. Chlorogonium spec.) were completely digested without transferring their chloroplasts.
The ciliation of Histiobalantium natans viridis is very characteristic. Between the dense fringe of cilia, elongated tactile cilia protrude in distances, which are 3–4 times longer and have a bristle-like character. They are called tactile cilia and are project radially in resting specimens.
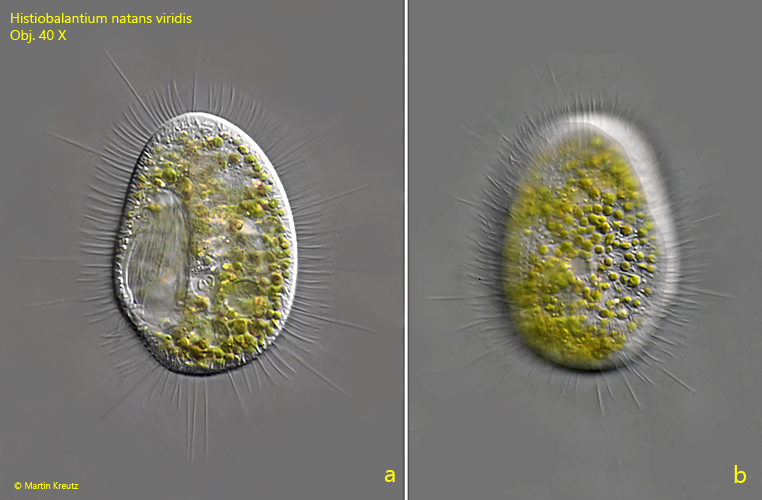
Fig. 1 a-b: Histiobalantium natans viridis. L = 84 µm. A resting specimen with spread tactile cilia from ventral (a) and from dorsal (b). Obj. 40 X.
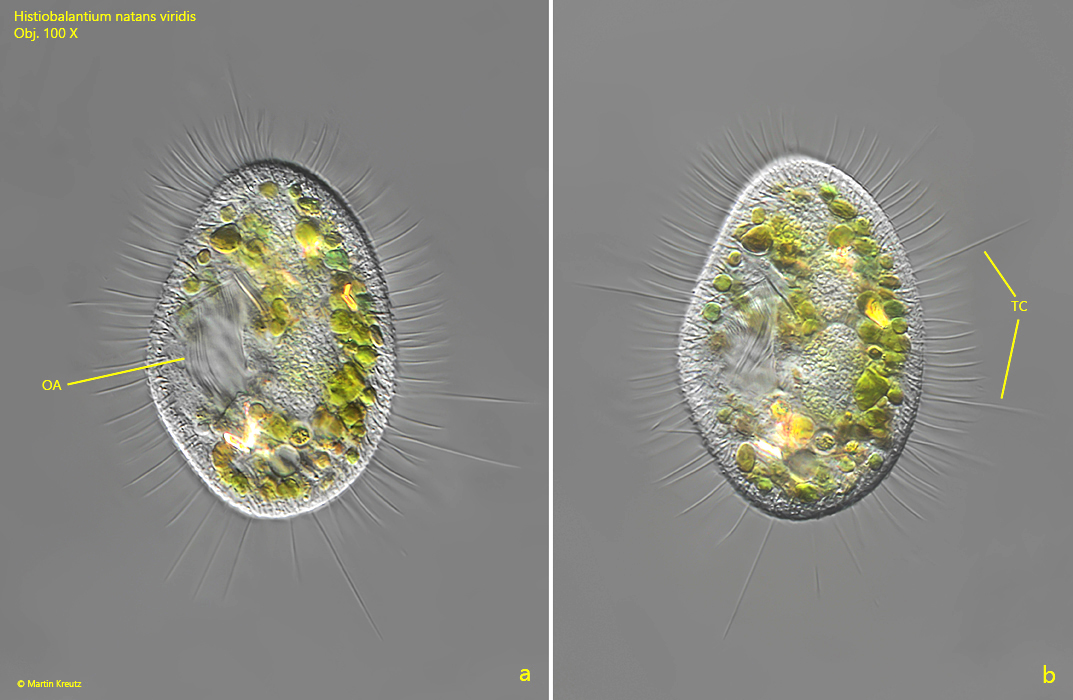
Fig. 2 a-b: Histiobalantium natans viridis. L = 62 µm. Two focal planes of a second specimen from ventral. OA = oral apparatus, TC = tactile cilia. Obj. 100 X.
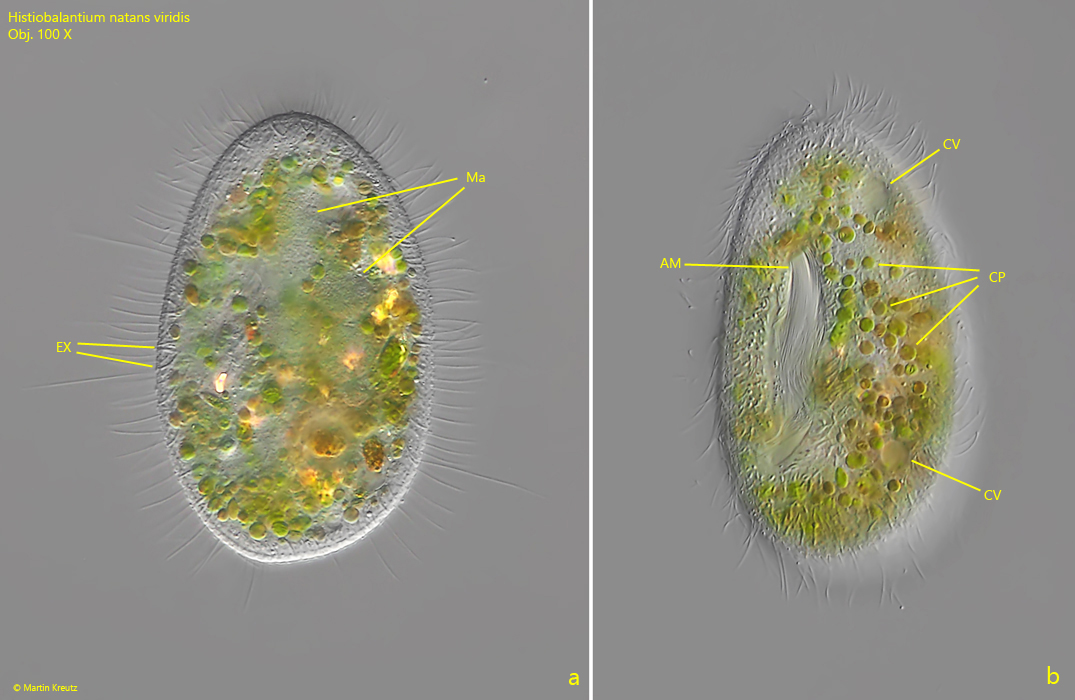
Fig. 3 a-b: Histiobalantium natans viridis. L = 55 µm. Two focal planes of a third specimen from ventral. Note the ingested cleptoplasts (CP) scattered in the cytoplasm. AM = adoral membranelle, CV = contractile vacuoles, Ma = macronucleus (consisting of two macronuclear nodules), EX = extrusomes. Obj. 100 X.
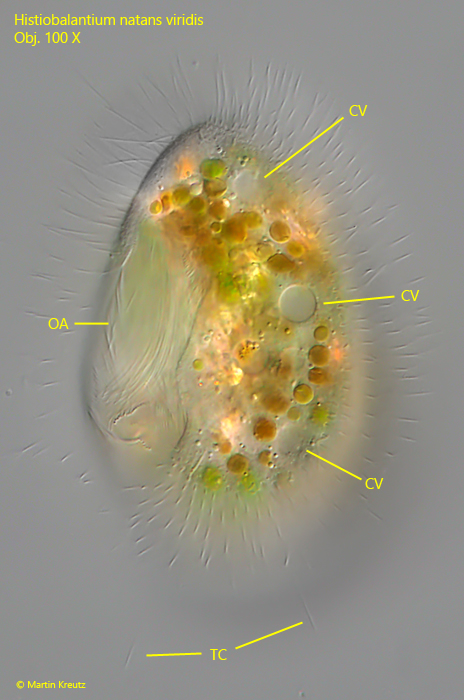
Fig. 4: Histiobalantium natans viridis. L = 72 µm. A specimen from ventral with focal plane on the row of contractile vacuoles (CV). OA = oral apparatus, TC = tactile cilia. Obj. 100 X.
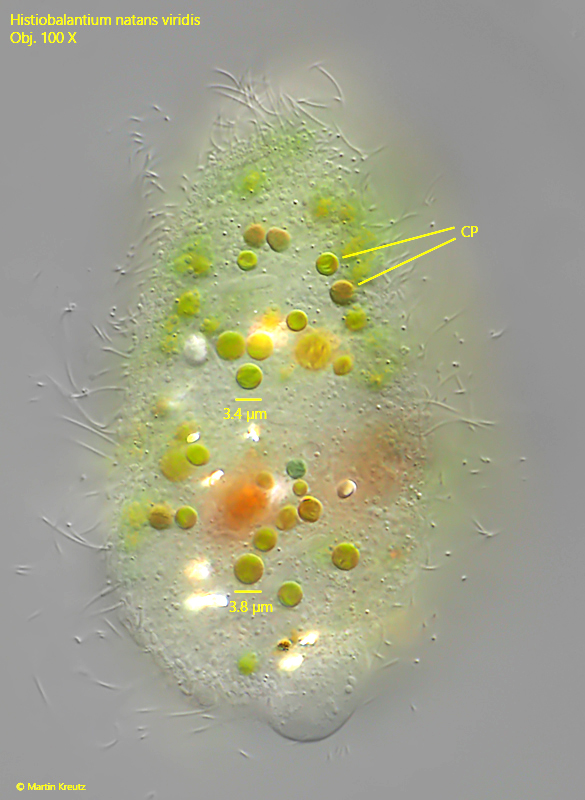
Fig. 5: Histiobalantium natans viridis. A slightly squashed specimen from dorsal with focal plane on the ingested cleptoplasts (CP). Obj. 100 X.
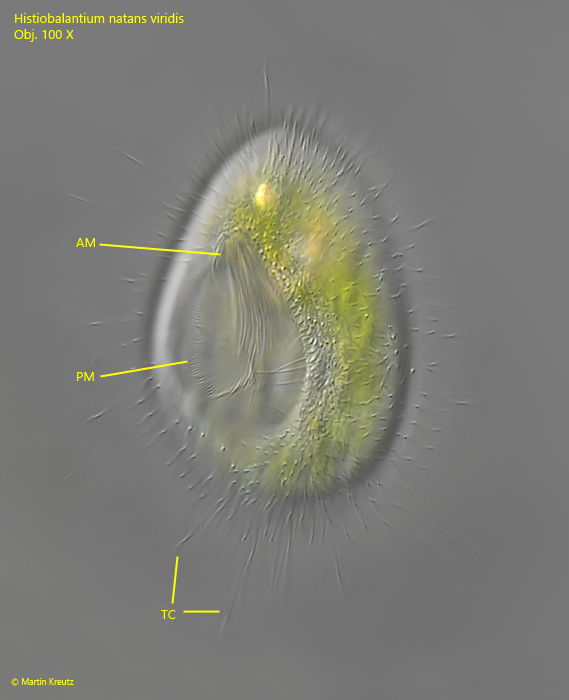
Fig. 6: Histiobalantium natans viridis. L = 67 µm. A resting specimen from ventral with the prominent oral apparatus. AM = adoral membranelle, PM = paroral membrane. TC = tactile cilia. Obj. 100 X.
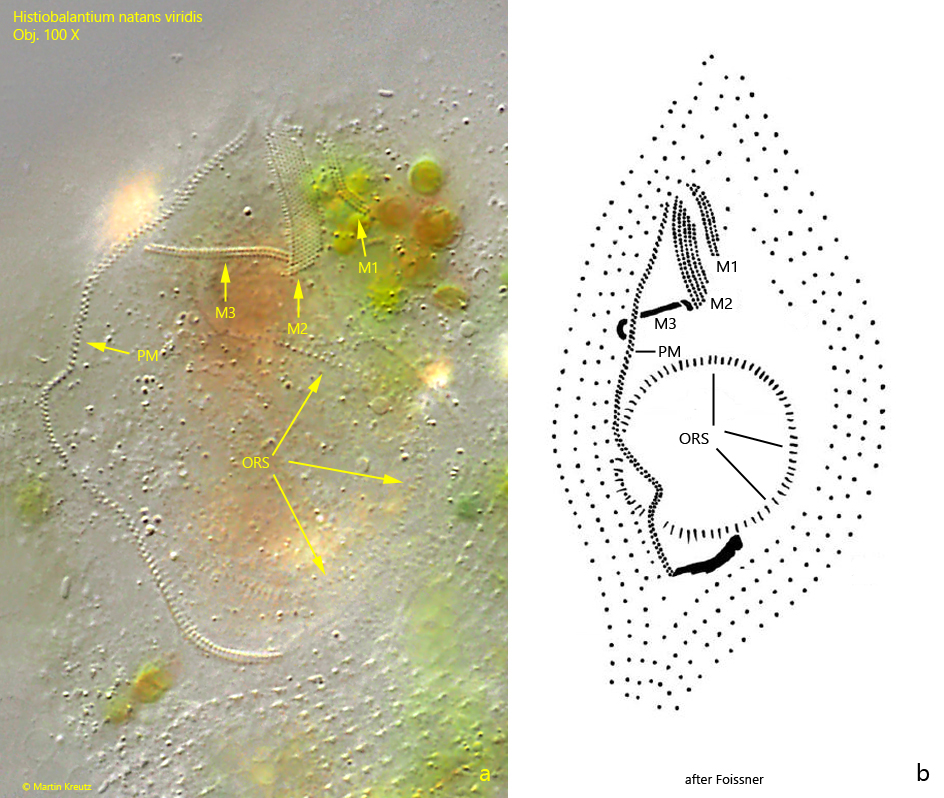
Fig. 7 a-b: Histiobalantium natans viridis. The oral apparatus in the strongly squashed specimen (a) and a schematic drawing of it for comparison (b). Note the three adoral membranelles (M1 – M3) and the curved course of the paroral membrane (PM). Also the almost circular oral rod structure (ORS) is visible. Obj. 100 X.
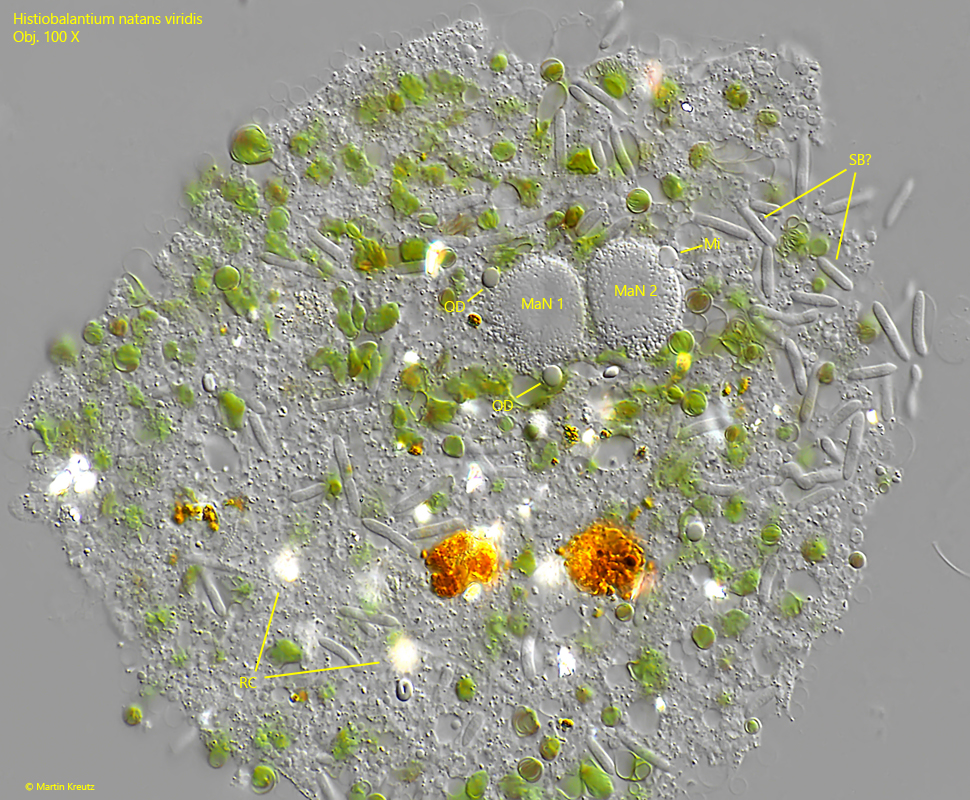
Fig. 8: Histiobalantium natans viridis. In this strongly squashed specimen the two macronuclear nodules (MaN 1, MaN 2) are visible and one of the two micronuclei (Mi). Also a large amount of bacteria is visible. Probably symbiotic bacteria (SB?) because all of them are identical. OD = oil droplets, RC = refractive crystals. OD = oil droplets, RC = refractive crystals. Obj. 100 X.
I could observe the form Histiobalantium natans nigricans (s. drawing 3 above) frequently between 2008 and 2014. After that the findings have become much rarer. You can recognize Histiobalantium natans nigricans by the slightly bean-shaped form and by the fact that the dense concentration of cleptoplasts is missing. Instead, there are many reserve grains in the cytoplasm, especially in the posterior end. In DIC, these reserve grains glow brightly, while in brightfield illumination they appear black. I assume that the naming of Kahl is based on this effect. The form Histiobalantium natans nigricans is also longer than the stockier form Histiobalantium natans viridis. In my population the specimens of Histiobalantium natans nigricans were always longer than 80 µm (s. figs. 9 a-b and 10 a-b).
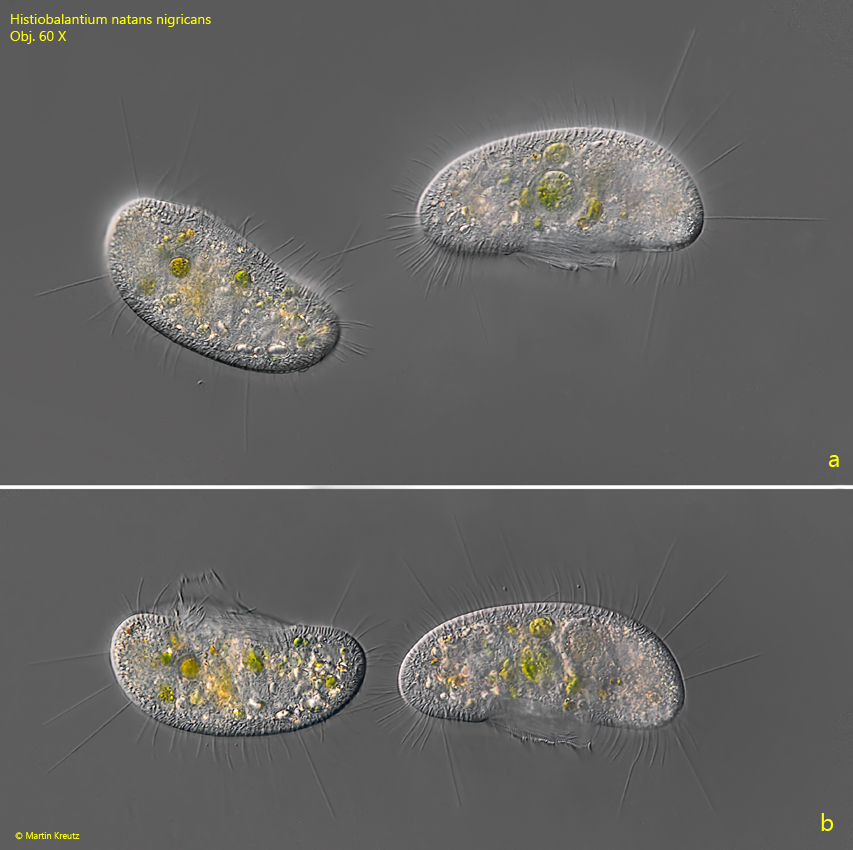
Fig. 9 a-b: Histiobalantium natans nigricans. 83–94 µm. A pair of freely swimming specimens. Specimens of this form are slightly bean-shaped. Obj. 60 X.
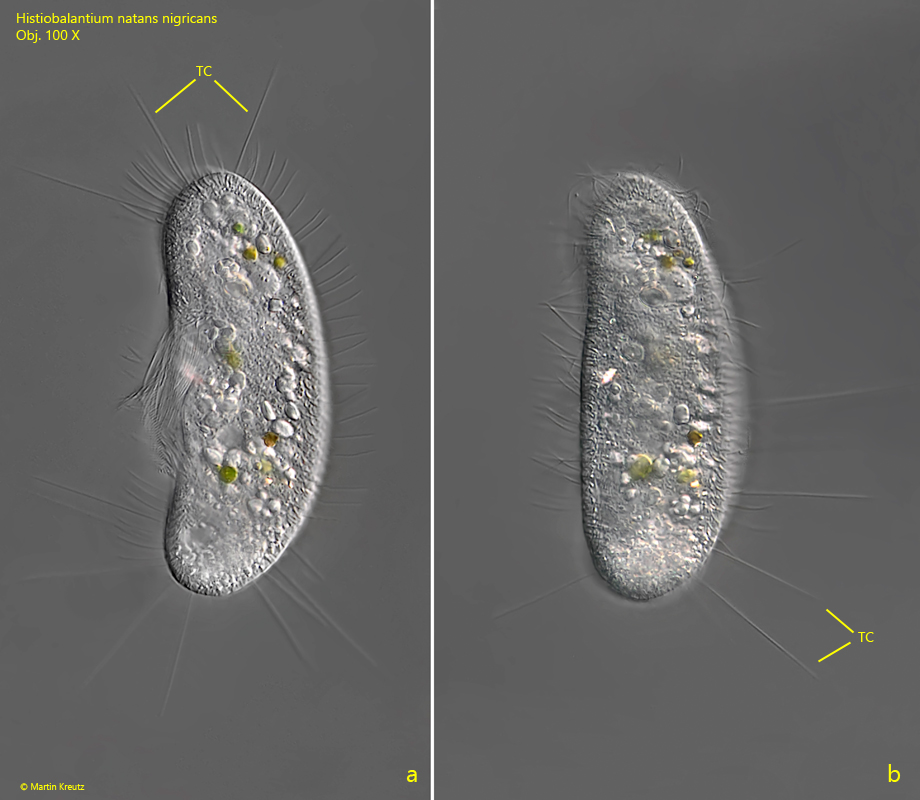
Fig. 10 a-b: Histiobalantium natans nigricans. 97 µm. A resting specimen with spread tactile cilia (TC). Obj. 100 X.