Metopus gibbus Kahl, 1927
Most likely ID: n.a.
Synonym: n. a.
Sampling location: Ulmisried, Simmelried
Phylogenetic tree: Metopus gibbus
Diagnosis:
- body pyriform, preoral dome hemispherical
- length 30–55 µm
- reniform macronucleus in preoral dome
- five perizonal rows of cilia
- adoral zone reaches to posterior third
- contractile vacuole terminal
- synciliary caudal cilia
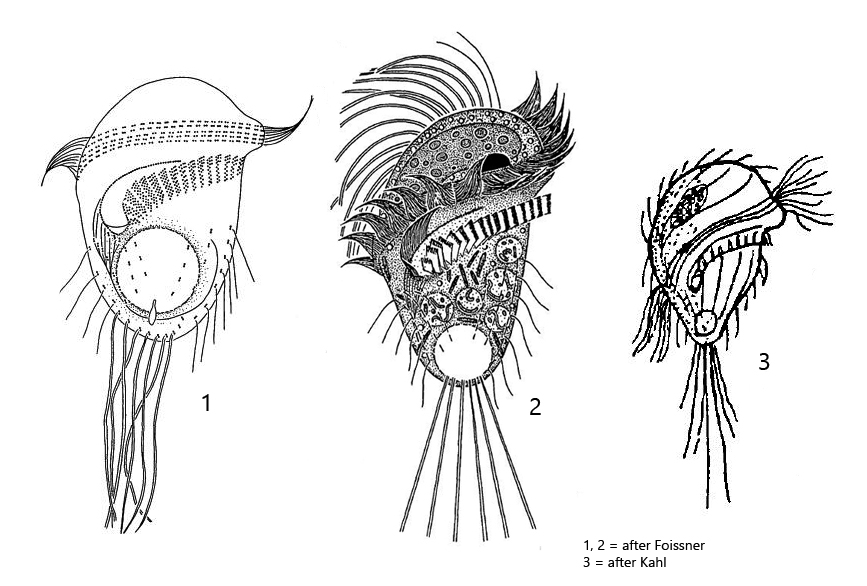
I find Metopus gibbus regularly, but rarely. The species was first described by Kahl, who, however, found only a few specimens and therefore had difficulties to provide a detailed description. After that Foissner, Agatha and Berger (2002, s. Literature) provided a redescription of Metopus gibbus in 2002 (s. Litertaure). For this purpose, populations from Croatia, Brazil and Namibia were examined.
The specimens of my population were between 25–36 long and thus correspond to the size specification of Kahl, who states about 30 µm. The specimens studied by Foissner et al. averaged 40 µm in length. The adoral zone of my specimens ran almost across the cell equator (s. figs. 1 a and 2 a) not slightly obliquely as drawn by Kahl and Foissner (s. drawings above). Both authors mention the long caudal cilia, which are drawn behind like a tail when swimming. These are synciliary cilia, where 2–3 cilia each stick together (s. fig. 3). In fact, the specimens in my population had synciliary caudal cilia up to 70 µm long (s. fig. 2 a). Foissner et al. give a length of 30–35 µm for them. On some specimens I could detect bacteria adhering to the pellicle (s. figs. 1d and 2a). However, Foissner et al. describe a granulated pellicle, where the granules are supposed to be ellipsoidally shaped and therefore resemble bacteria. Possibly the adhering bacteria are therefore the described granules, whereby it was clearly recognizable for me that these bacteria adhered to the pellicle and were not located under the pellicle. I only checked the shape of the macronucleus on one specimen (s. fig. 5). In my specimen, it was distinctly spherical and not kidney-shaped as described by Foissner et al. I also could not detect any bacteria in the macronucleus, but many rods in the cytoplasm, which could be symbiotic bacteria (s. fig. 5). So there are some differences to the redescription by Foissner et al. However, because of the small size, the very long synciliary caudal cilia and the position and shape of the adoral zone I do not see any determination alternative to Metopus gibbus.
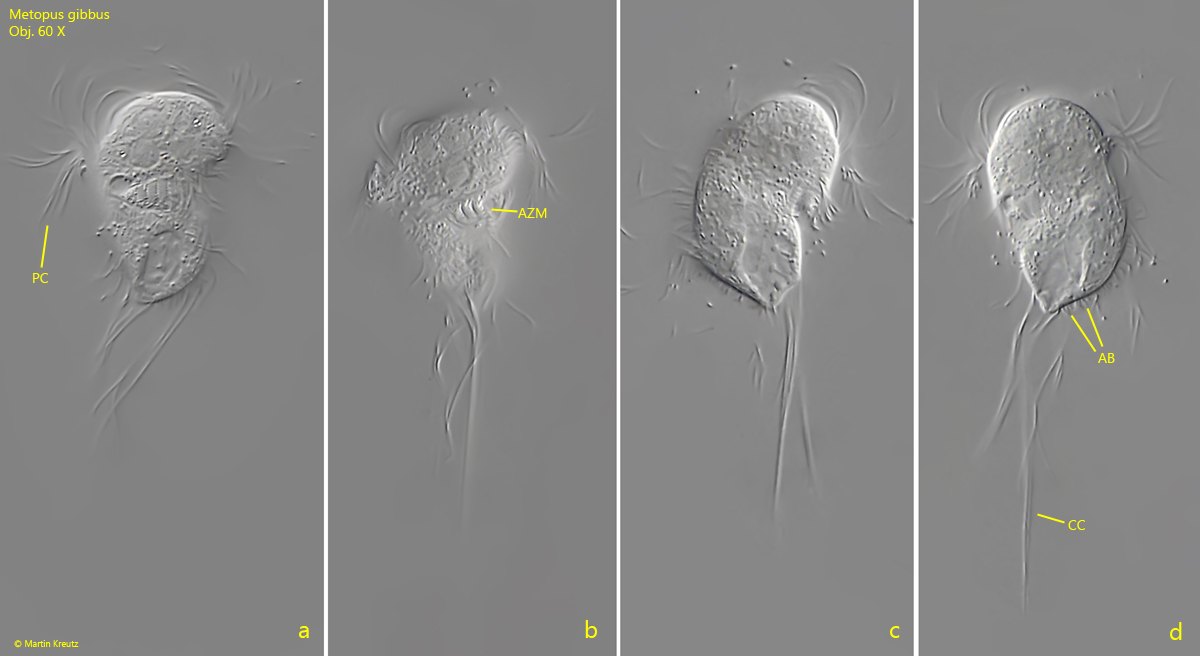
Fig. 1 a-d: Metopus gibbus. L = 34 µm. A freely swimming specimen from ventral (a, b), right (c) and left (d). AB = adhering bacteria on pellicle, AZM = adoral zone of membranelles, CC = caudal cilia, PC = perizonal cilia. Obj. 60 X.
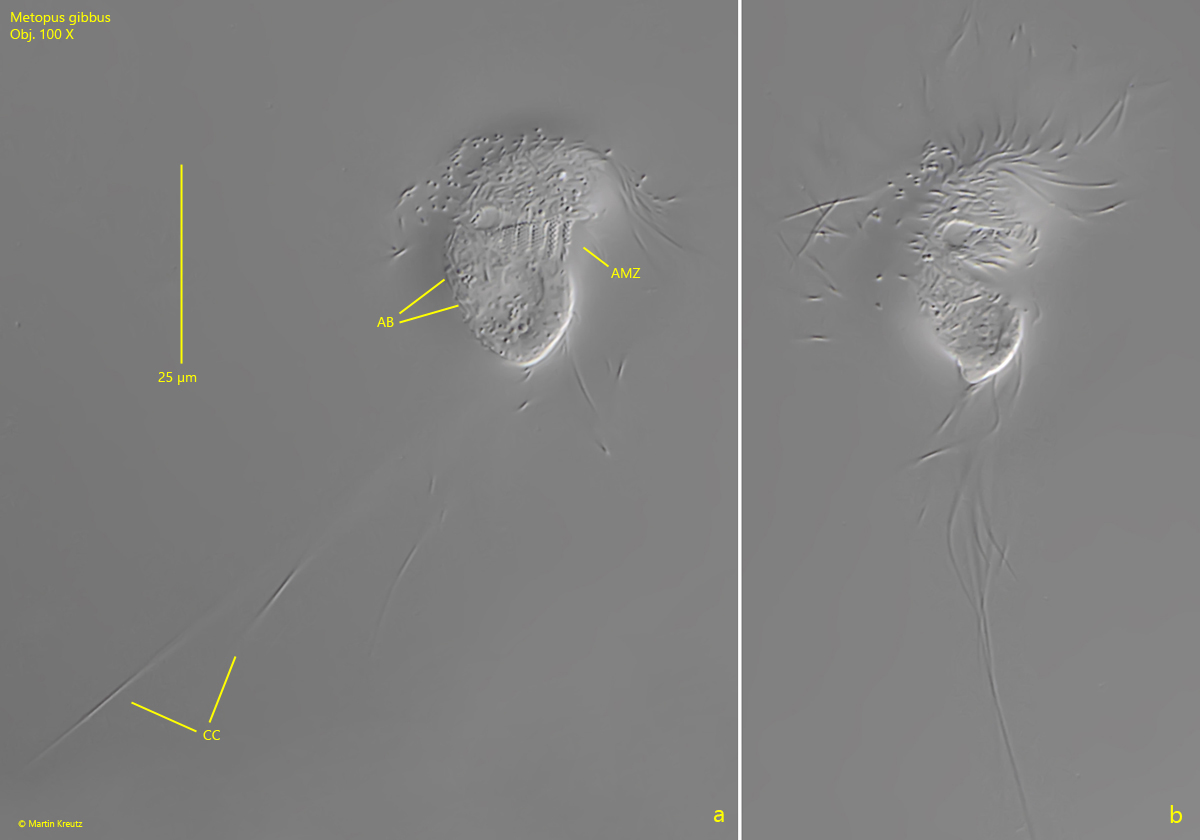
Fig. 2 a-c: Metopus gibbus. L = 26 µm. A second, freely swimming specimen from ventral. Note the very long caudal cilia (CC) with a length of 70 µm. AB = adhering bacteria on pellicle, AZM = adoral zone of membranelles. Obj. 100 X.
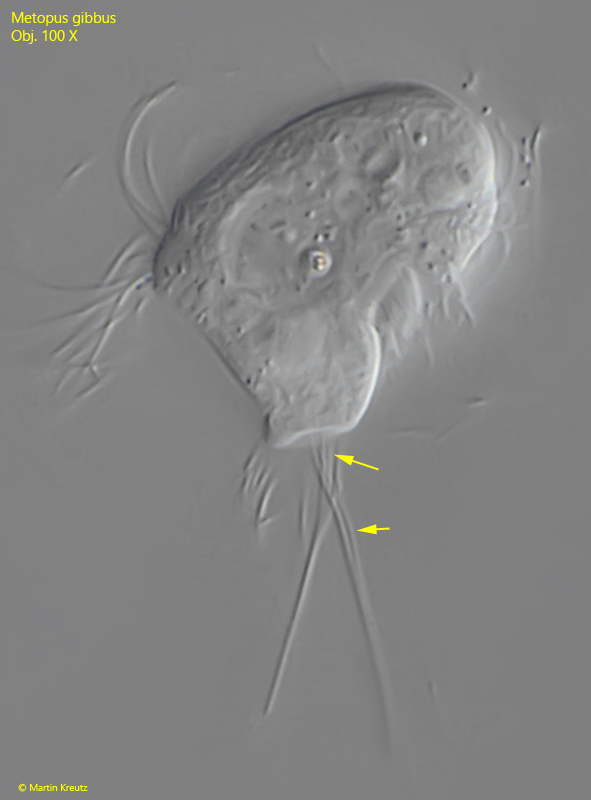
Fig. 3: Metopus gibbus. L = 25 µm. A slightly squashed specimen with a strongly overhanging dome and a truncated posterior end. Note that 2–3 cilia each form the caudal syncilia (arrows). Obj. 100 X.
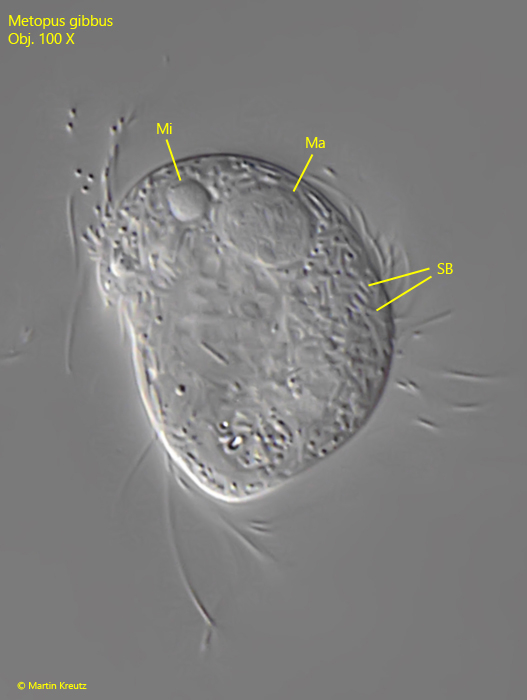
Fig. 5: Metopus gibbus. The spherical macronucleus (Ma) and the adjacent micronucleus (Mi) in a strongly squashed specimen. The cytoplasm seems to be filled with symbiotic bacteria (SB). Obj. 100 X.