Nassulopsis elegans
(Ehrenberg 1834) Foissner, Berger & Kohmann, 1994
Most likely ID: n.a.
Synonym: Nassula elegans
Sampling location: Kaltbrunn pond
Phylogenetic tree: Nassulopsis elegans
Diagnosis:
- length about 120–250 µm
- body pyriformed, flexible, often anterior third is bent backwards
- synhymenium present with a dorsal gap
- oral basket in anterior sixth, consisting of 24–40 rods
- ventral row of 3–6 contractile vacuoles
- macronucleus ellipsoidal with 5–25 micronuclei
- distinct pigment spot located dorsally at the anterior end
- food vacuoles often intensely colored (by ingested cyanobacteria)
- extrusomes (Mucocysts) inconspicuous
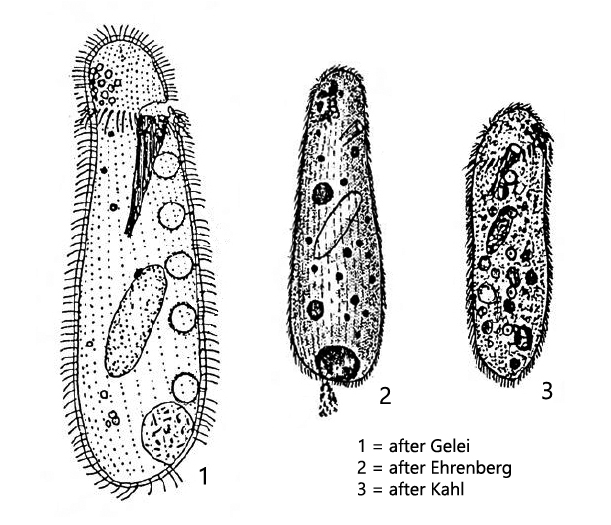
I found Nassulopsis elegans in 1996 in a small, shady forest pond near Kaltbrunn, about 6 km northwest of Constance. There I noticed mats of cyanobacteria floating on the water, almost black in color. I skimmed these off and found in them a large population of Nassulopsis elegans, which feed mainly on cyanobacteria.
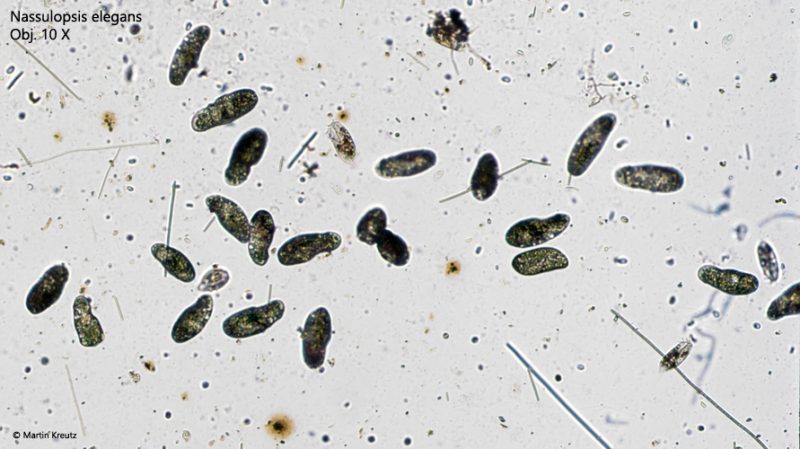
Fig. 1: Nassulopsis elegans. Overview of a sample with Nassulopsis elegans grazing on filamentous cyanobacteria (likely Oscillatoria). Obj. 10 X.
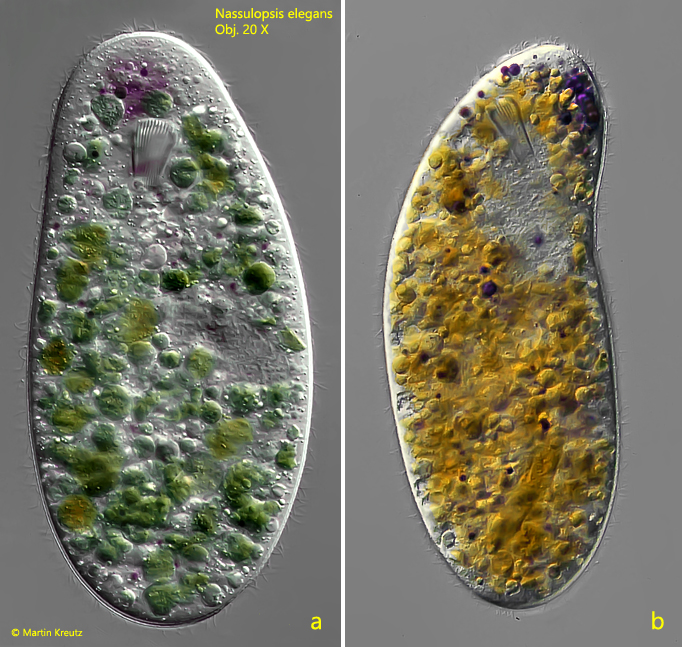
Fig. 2 a-b: Nassulopsis elegans. L = 240 µm. Slightly squashed specimen in ventral view (a) and a second specimen in lateral view from left (b). Note the purple pigment spot in the “neck”, consisting of intense purple colored droplets. Obj. 20 X.
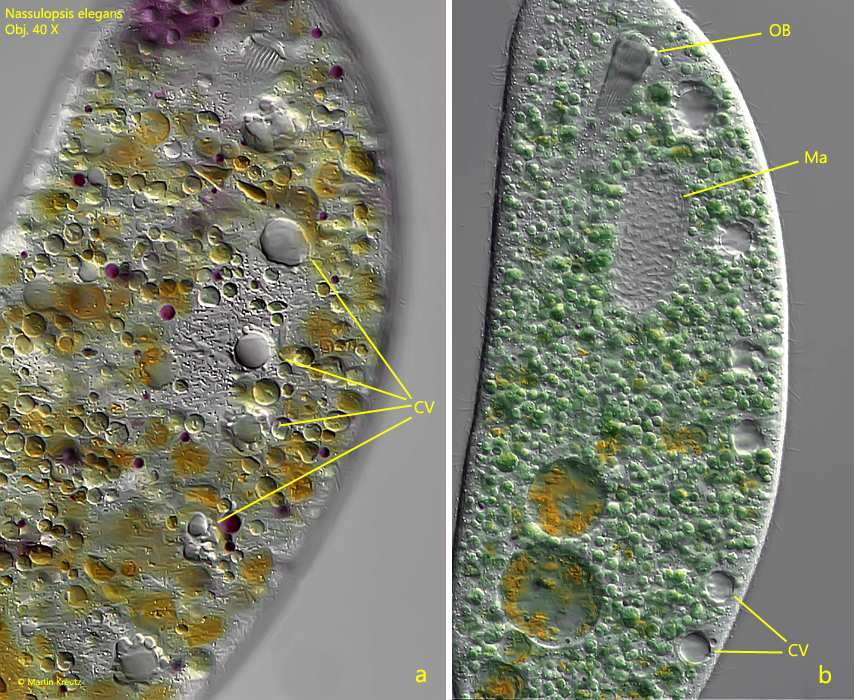
Fig. 3 a-b: Nassulopsis elegans. L = 240 µm. The ventral row of 6 contractile vacuoles in two different specimens. a) ventral view, b) lateral view from right. CV = contractile vacuoles, Ma = macronucleus, OB = oral basket. Obj. 40 X.
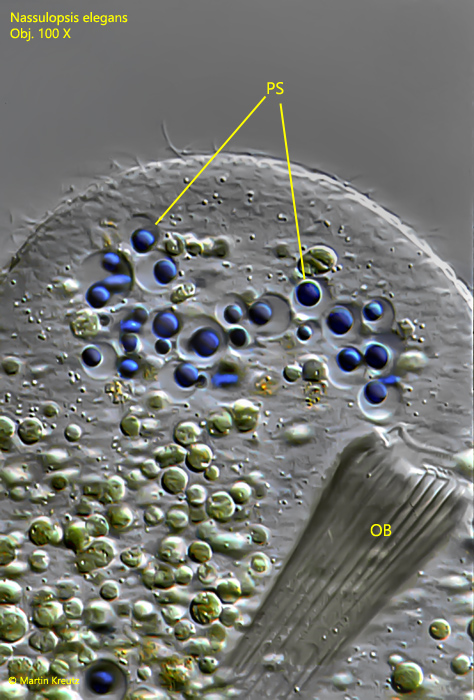
Fig. 4: Nassulopsis elegans. Detailed view of the dorsally located pigment spot. The intensely colored droplets are enclosed in separate vacuoles. OB = oral basket, PS = pigment spot. Obj. 100 X.
If a specimen of Nassulopsis elegans is squashed under the coverslip and the microscope focused on the pellicle, a honeycomb-like pattern of alveoli is revealed. In each of the alveoli a mucocyst (MU) is visible. The mucocysts can enlarge explosively (e.g. by mechanical stress or an attack by a predator) and form a layer of mucus around the ciliate.

Fig. 5: Nassulopsis elegans. Detailed view of the pellicle with a honeycomb-like pattern of alveoli. In each of the alveoli a mucocyst (MU) is located. Obj. 100 X.
Many ciliates with a oral basket are adapted to a specific diet. This is also true for Nassulopsis elegans, which feeds exclusively on filamentous cyanobacteria of the genera Oscillatoria and Phormidium. The ciliate can reliably distinguish these cyanobacteria from other filamentous blue-green algae. This can be easily demonstrated by feeding with cyanobacteria of other genera. Other cyanobacterial genera are not accepted even when the specimens are completely starved (free of food vacuoles). The oral basked of Nassulopsis elegans consisting of 24–40 rods (nemadesmata) and is ideally adapted to the ingestion of cyanobacterial filaments. In the specimens of the population from Kaltbrunn the number of rods was consistently 24–26. The rods are movable against each other and the basket can change the diameter adapting to the exact kind of ingested cyanobacterium. During feeding, the rods tightly enclose the filamentous cyanobacterium. I could observe the feeding process of Nassulopsis elegans very frequently, but rarely with the coverslip in place. In one of these few cases, however, I was able to document the process accurately. At first Nassulopsis elegans is gliding along the cyanobacteria filaments with the oral basket in contact to the filament to find an end. Sometimes, however, phagocytosis already began without finding the end of the filament. In these cases, the filament was kinked and the resulting double filament was engulfed. In cases where the end of the filament was found, the basket was placed over it and the rather spectacular uptake of the cyanobacterial filament began (s. fig. 6).

Fig. 6: Nassulopsis elegans. The feeding process of a filamentous cyanobacterium in time intervals. Obj. 20 X.
I could determine the speed at which the cyanobacteria moves through the oral basket from various observations to be approximately 5 µm/sec. This value seems to be independent of the diameter of the cyanobacterial filament and the individual specimen. While the cyanobacterial filament is gliding through the oral basket, no movement of the oral basket or activity around the oral basket can be detected light-microscopically, since the directed force is generated at the molecular level. However, the forces acting there eventually become obvious when the cyanobacterial filament has reached the posterior end of the cell (s. fig. 6, after 5 sec). 1). Now some very amazing things happen. The ingestion process will be not interrupted. Instead, the cyanobacteria bend through. The pellicle is stretched in a “bow” (s. fig. 6, after 15 sec), proving its extraordinary elasticity and tensile strength. Due to the resistance, the oral basket now protrudes like a snout. A short time later, the cyanobacteria break (s. fig. 6, after 15 sec) and are folded subsequently in the posterior end of Nassulopsis elegans. In the process, the ciliate sometimes assumes bizarre shapes. Without the coverslip pressure, the filament can also fold three-dimensionally what can cause tetrahedrally shaped individuals. After about 4 min the ingestion process stops what can be seen by a clear slowing down of the uptake. The part of the cyanobacterial filament, which already passed the basket, is partially digested by supplied lysosomes and finally the filament is cut by this enzymatic activity.
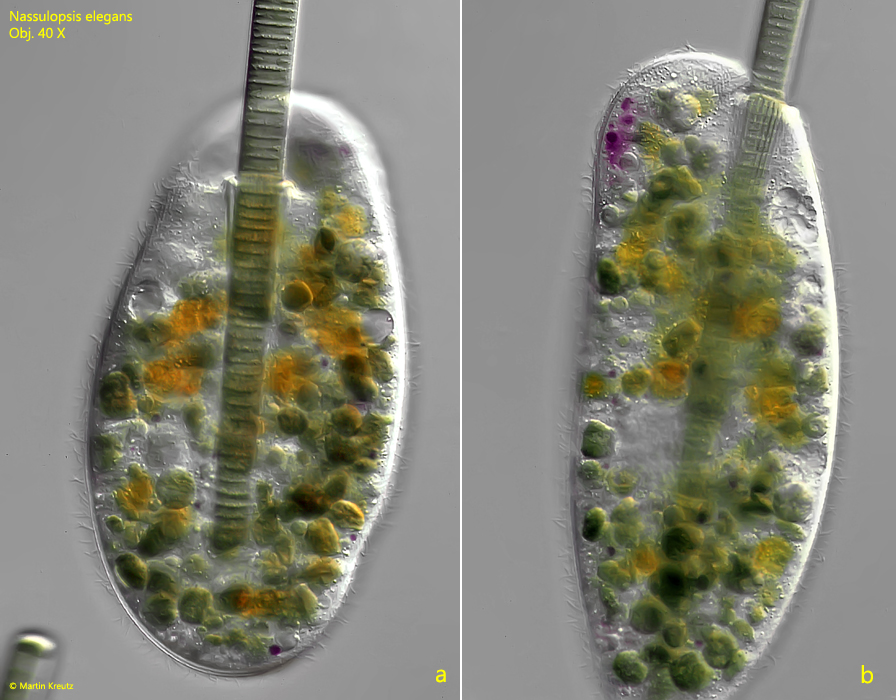
Fig. 7 a-b: Nassulopsis elegans. Detailed view of the feeding process in ventral view (a) and lateral view from right (b). Obj. 40 X.
During the passage of the oral basket, the ingested cyanobacterial filament is continuously enveloped by a membrane, which at first is tightly attached to the cyanobacterium. Just one minute later, the membrane begins to detach to form an elongated vacuole (s. fig. 8). The increase in volume of the food vacuole most likely occurs through fusion with lysosomes from the plasma, which bring in further digestive enzymes. After only two minutes, the lumen of this “giant vacuole” turns distinctly purple. Seconds later, the disintegration of the filamentous cyanobacterium begins.
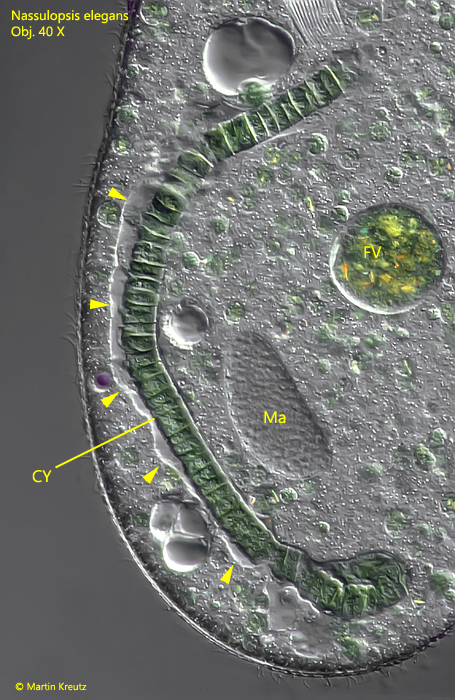
Fig. 8: Nassulopsis elegans. Ingested cyanobacterial filament in an elongated vacuole. The membrane of the vacuole is detached from the cyanobacterium (arrowheads). CY = filamentous cyanobacterium, FV = food vacuole, Ma = macronucleus. Obj. 40 X.
In the Petri dishes with the cultures of Nassulopsis elegans I could make further observations of the life cycle of this ciliate. With good feeding of cyanobacteria, I was able to observe numerous cell divisions. First visible sign for the beginning of cell division is an incipient constriction of the cell center. In parallel to this the oral basket has to double. In Nassulopsis elegans, the old oral basket is degraded and then a new one is formed in each half of the cell. The new oral baskets are formed from previously linearly arranged microtubule components, which arrange themselves into a circle as differentiation progresses (s. Fig. f).
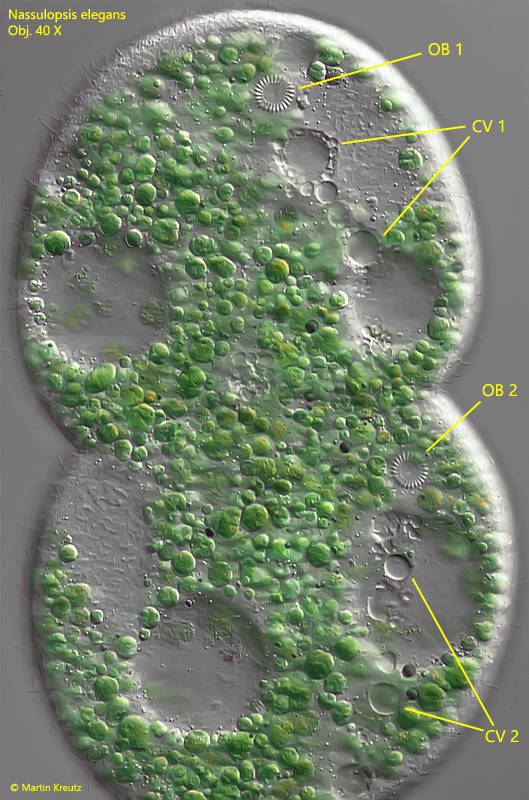
Fig. 9: Nassulopsis elegans. A specimen during cell division. The oral baskets are already doubled (OB 1 and OB 2) as well as the rows of contractile vacuoles (CV 1 and CV 2). Obj. 40 X.
Sometimes the construction of the new oral basketes in the two new cells is faulty. This results in individuals which carry 2, 3 or even more oral baskets. I could observe individuals with 3 oral baskets often (s. fig. 10). This is not unusual. The surplus baskets are degraded in the course of the life cycle.
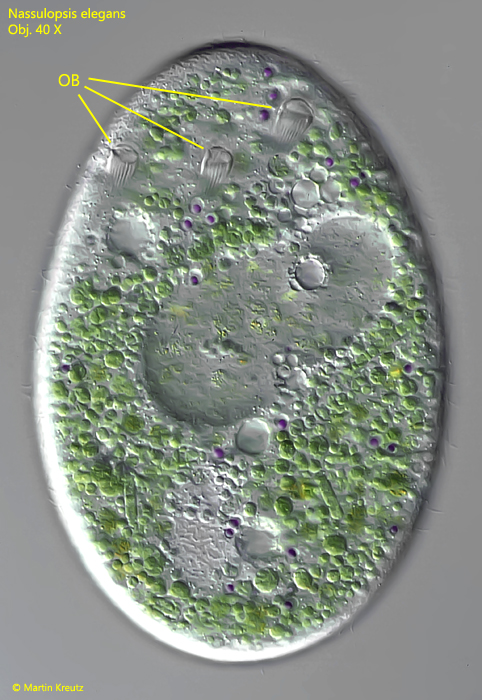
Fig. 10: Nassulopsis elegans. A specimen with 3 baskets caused by an incorrect cell division. OB = oral baskets. Obj. 40 X.
In the culture, sexual reproduction by conjugation could also be observed. However, I found only 2 conjugating pairs among hundreds of individuals. The conjugation of ciliates with an oral basket is particularly interesting, because the oral openings have to fuse during conjugation. It begins in Nassulopsis elegans with the formation of a bridge of cytoplasm between the oral baskets (s. fig. 11).
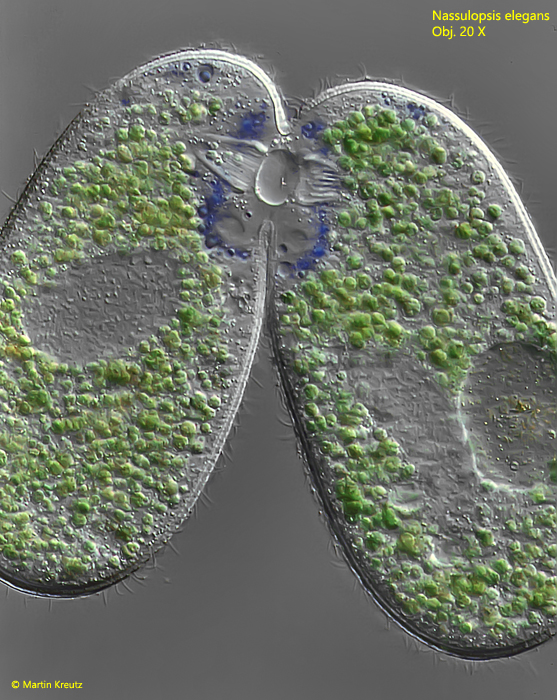
Fig. 11: Nassulopsis elegans. Two specimens connected via the oral baskets during conjugation. Obj. 20 X.
This cytoplasm bridge is very stable and cannot be loosened even by coverslip pressure. A vacuole-free area forms around the oral baskets. A large number of the colored vacuoles accumulate around the contact site. These were obviously transferred from the dorsally located pigmented spot. In a few specimens I could observe the meiotic reduction division. In these cases the micronuclei appear elongated. (s. fig. 12).
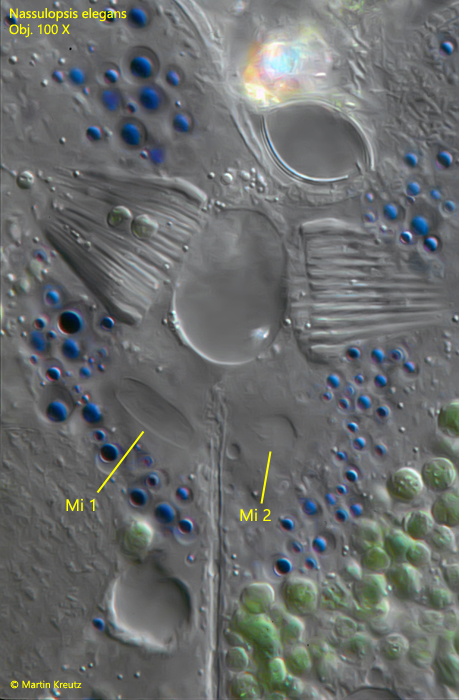
Fig. 12: Nassulopsis elegans. Detailed view of the contact site between two spesimens. Note the blue colored droplets arranged around the connecting site and the two micronuclei (Mi 1, Mi 2) in a meiotic division process. Obj. 100 X.
With regular feeding, a high population density of Nassulopsis elegans can be produced in microaquaria. If the feedings are then omitted, the specimens become increasingly transparent due to the lack of food vacuoles. Later, even the colored vacuoles at the dorsal anterior end are degraded. These starvation forms are conspicuous in their unusual shape. They are dented in various places, so that the former pyriform shape is difficult to recognize. About two days after cessation of feeding, mass encystment begins. Encystment is initiated by a marked slowing of movement. The specimens eventually encyst and appear to excrete a thin but firm mucus coat. This apparently forms the later cyst envelope. In the round cysts the oral basket is still clearly visible as well as a distinct orange or brownish spot (s. fig. 13 and 14).
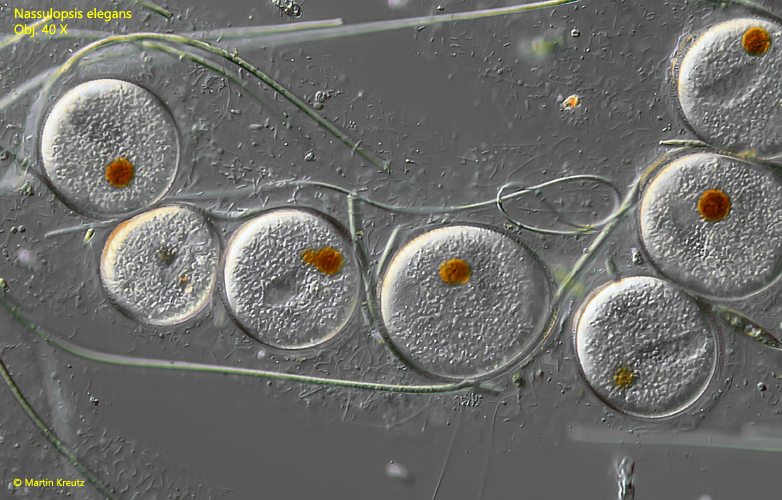
Fig. 13: Nassulopsis elegans. Accumulation of globular resting cysts with a distinct orange spot. Obj. 40 X.
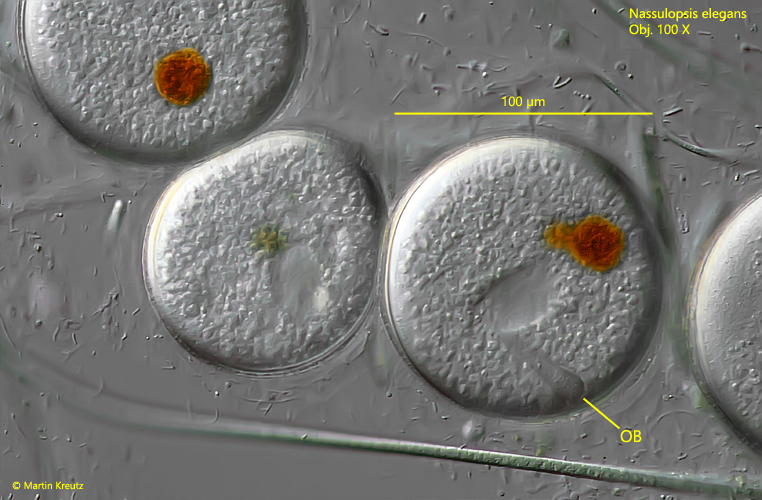
Fig. 14: Nassulopsis elegans. The globular cysts have a diameter of 80–105 µm. In some cysts the oral basket (OB) can be recognized. Obj. 100 X.