Paramecium bursaria
(Ehrenberg, 1831) Focke, 1836
Most likely ID: n.a.
Synonym: Chloroparamecium bursaria
Sampling location: Ulmisried, Simmelried, Bussenried, Bündtlisried, Purren pond
Phylogenetic tree: Paramecium bursaria
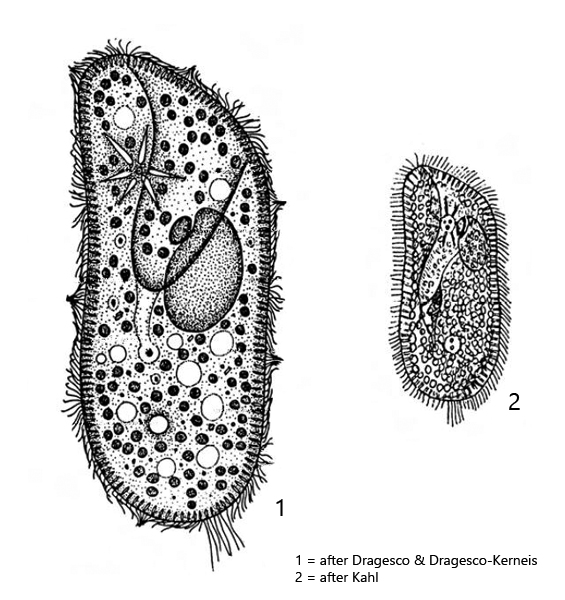
Diagnosis:
- length 85-150 µm, width 40-70 pm.
- cell foot-shaped, elliptical or ovate
- dorso-ventral flattened
- ventral oral groove near mid-cell, often indistinct
- macronucleus ellipsoid with one adjacent miconucleus
- extrusomes spindle-shaped, form a distinct fringe under the pellicle
- two contractile vacuoles with radial collecting canals
- each contractile vacuole with 2-3 excretion pori
- cells green due to symbiotic algae
- about 60 longitudinal rows of cilia form a median suture on the ventral side
- tuft of 10 – 17 µm long caudal cilia
Paramecium bursaria is a very common ciliate present in all my sites. It is found mainly in floating plant masses. In the sample vessels it collects at the surface on the side facing the light. Similar to Paramecium caudatum, it is one of the best studied ciliates ever. In particular, its symbiosis with algae of the genus Chlorella (s. fig. 9) has been intensively studied.
A reliable feature to distinguish Paramecium bursaria from the similar species Paramecium chlorelligerum are the caudalcilia. In Paramecium bursaria, they grow to a maximum length of 20 µm and are clearly spreaded (s. fig. 6). In addition, the two contractile vacuoles have collecting canals and no auxiliary vacuoles. The contractile vacuoles have also 2 – 3 clearly visible excretory pori (s. fig. 10). In Paramecium chlorelligerum it is only one porus.
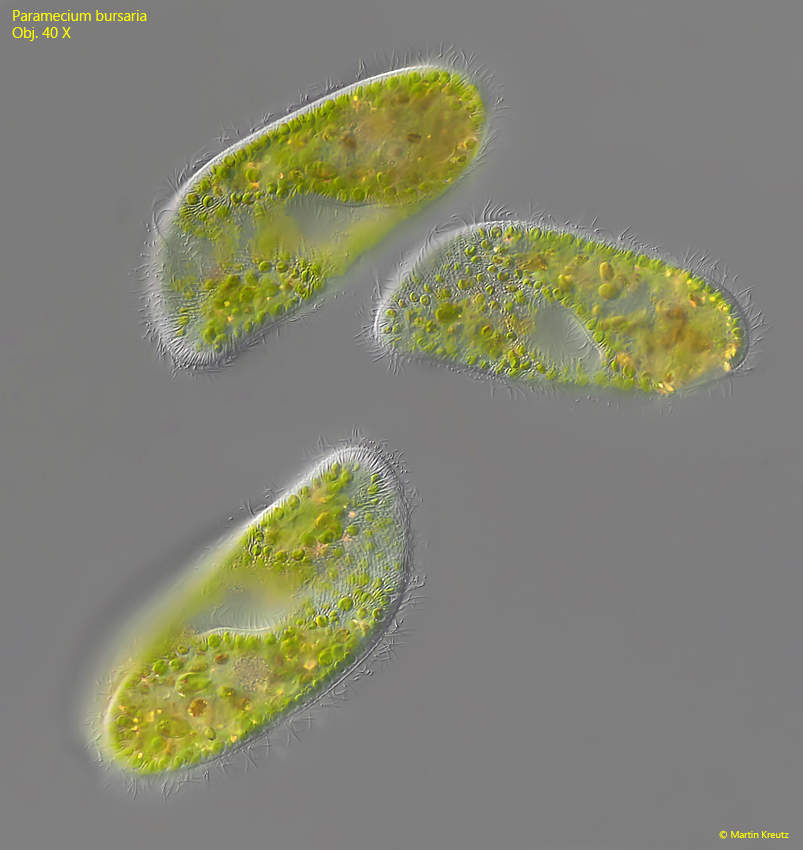
Fig. 1: Paramecium bursaria. L = 139 – 157 µm. Three freely swimming specimen. Obj. 40 X.
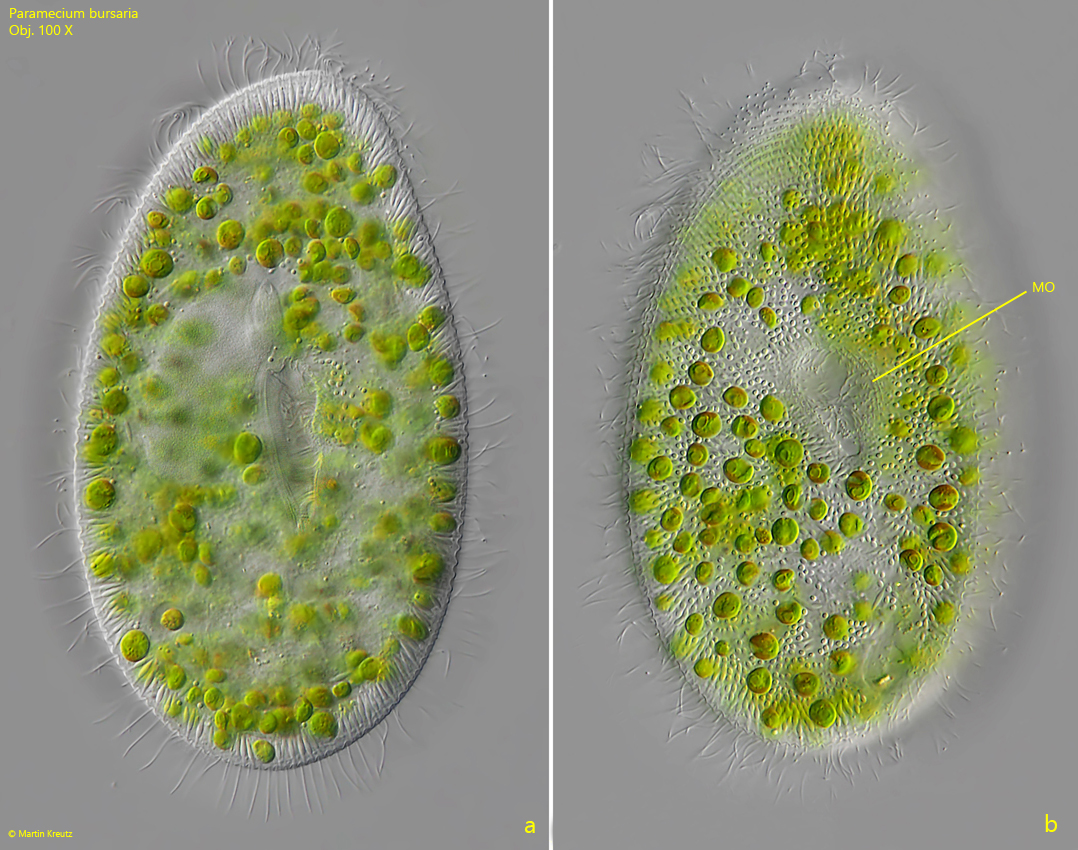
Fig. 2 a-b: Paramecium bursaria. L = 125 µm. Two focal planes of a slightly squashed specimen. Obj. 100 X.
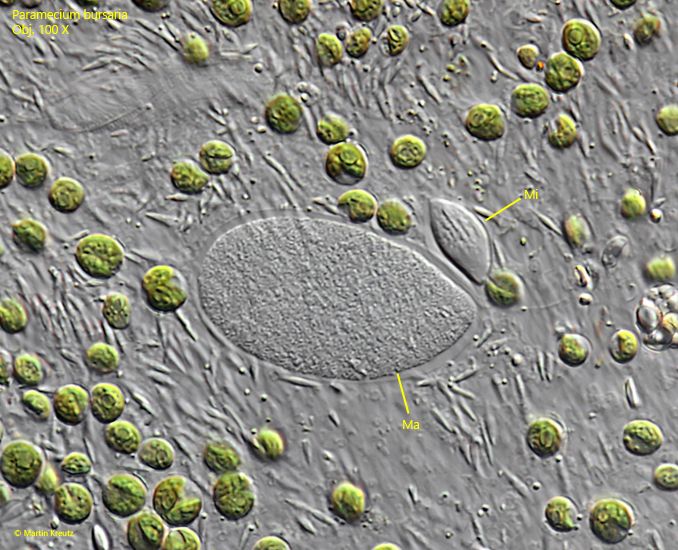
Fig. 3: Paramecium bursaria. The macronucleus (Ma) and the micronucleus (Mi) in a squashed specimen. Obj. 100 X.
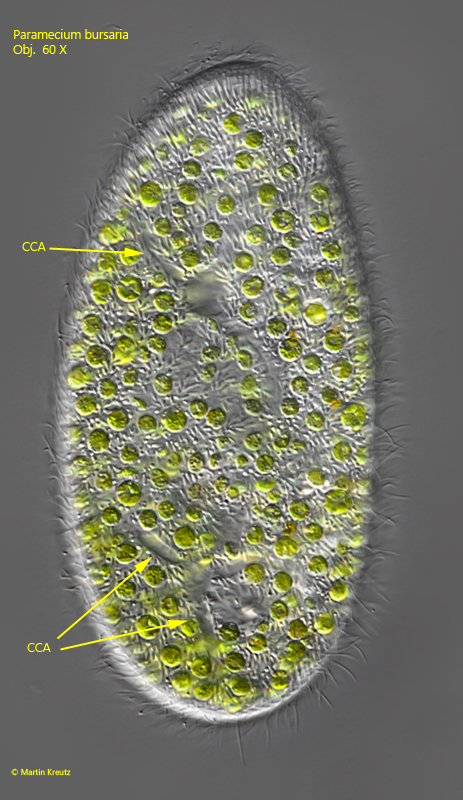
Fig. 4: Paramecium bursaria. L = 130 µm. The collecting channels (CCA) of the two contractile vacuoles. Obj. 60 X.
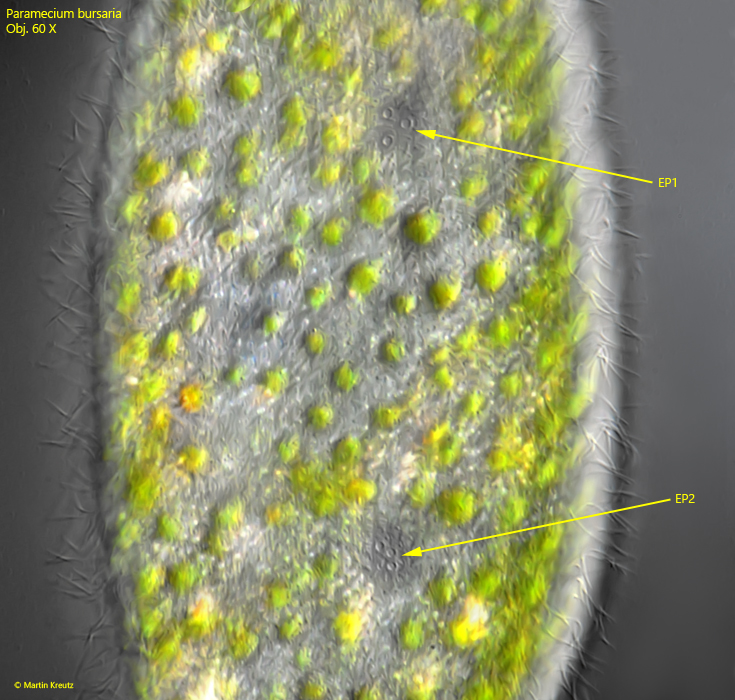
Fig. 5: Paramecium bursaria. The contractile vacuoles can have 2 or 3 excretion pori. This specimen has three excretion pori (EP1, EP2) for each contractile vacuole. Obj. 60 X.
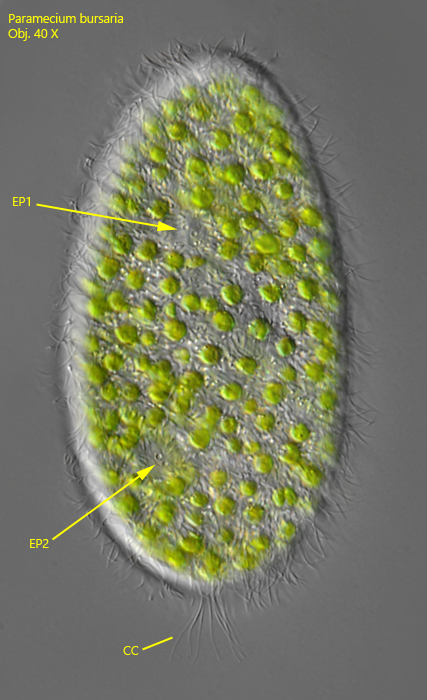
Fig. 6: Paramecium bursaria. L = 120 µm. The tuft of caudal cilia (CC) is located posterior on the dorsal side. The caudal cilia of this specimen are 17 µm long. The excretion pori (EP1, EP2) of the dorsal side are also in focus. Obj. 40 X.
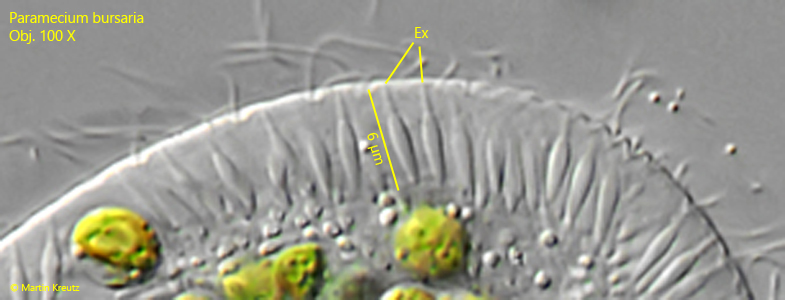
Fig. 7: Paramecium bursaria. The extrusomes (EX) are amphora-shaped and 5-6 µm long. Obj. 100 X.

Fig. 8: Paramecium bursaria. The ejected extrusomes are spear-shaped and 29 µm long. Obj. 100 X.

Fig. 9: Paramecium bursaria. The symbiotic algae of the genus Chlorella in detail. The single chloroplast in the cells contains one pyrenoid. The diameter of the spherical cells is 5–6 µm. Note the two symbiotic alage near the center of the image in cell division. Obj. 100 X.
In May 2021, I found a population of Paramecium bursaria in Simmelried with a large number of specimens in the process of conjugation. This process serves to exchange genetic material (stored in the micronuclei) and is associated with a prior, meiotic division of the diploid genetic material before the resulting haploid micronuclei of the conjugants can be exchanged. Because I had a large number of conjugants available, I was able to observe snapshot of the entire process in each, which otherwise takes about 20 hours. Interpretation of these snapshots is difficult because there are only few publications of the structures actually visible microscopically. However, a realistic graphical representation has been given by G. N. Calkins (s. fig. 10), who studied the processes during conjugation on Paramecium aurelia.
During meiosis, the diploid micronucleus first undergoes a twofold meiotic division resulting in 4 haploid nuclei, 3 of which are resolved. The remaining haploid nucleus divides again meitotically into a resting nucleus (remains in the mother cell) and a migrating nucleus (is exchanged with partner cell). Then both cells exchange their haploid migratory nuclei. After the exchange, the two exchanged migratory nuclei fuse with the resting nuclei remaining in the mother cells, resulting in a newly mixed diploid micronucleus in each of the two cells. This process was observed by Calkins in Paramecium aurelia as shown in figs. 10 a-e.
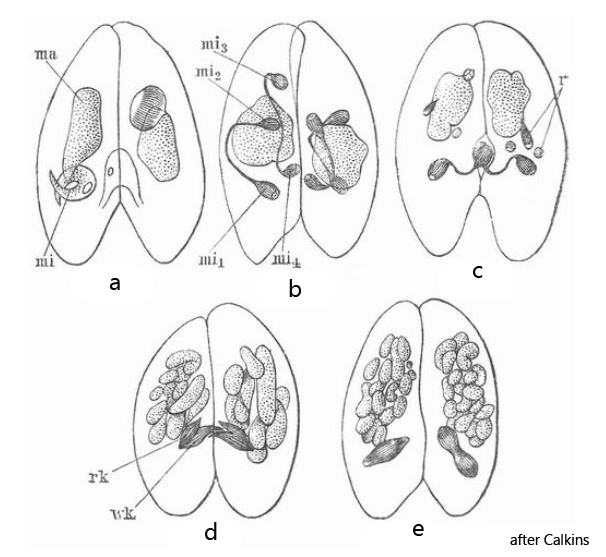
Fig. 10: The drawings of G. N. Calkins showing the actually visible process during the meiotic division of the micronuclei prior the exchange of genetic material during conjugation of Paramecium aurelia. Obj. 100 X.
The captions from “Meyer’s Großes Konversations-Lexikon” for the drawings by G.N. Calkins are as follows:
- Fig. 10a: two ciliates placed side by side for fertilization with main nucleus ma and secondary nucleus mi.
- Fig. 10b: division of the minor nucleus mi into four small minor nuclei.
- Fig. 10c: the last remains (r) of three minor nuclei. Formation of a migrating nucleus (wk) and a resting nucleus (rk) from the fourth minor nucleus.
- Fig. 10d: cross-fertilization by the migrating nuclei.
- Fig. 10e: division processes on the product of the fusion of the resting nucleus with the newly added migrating nucleus.
Based on the description and the drawings of Calkins, I have now tried to interpret my own observations.
The conjugation begins with the establishment of a plasma bridge between the mouth regions of the two conjugants (s. fig. 11). For this purpose, both cells temporarily swim in parallel and remain attached to each other with their cilia (this is how I have seen it). Once this initial contact is established, the mouth regions begin to fuse, forming a permanently bound pair.
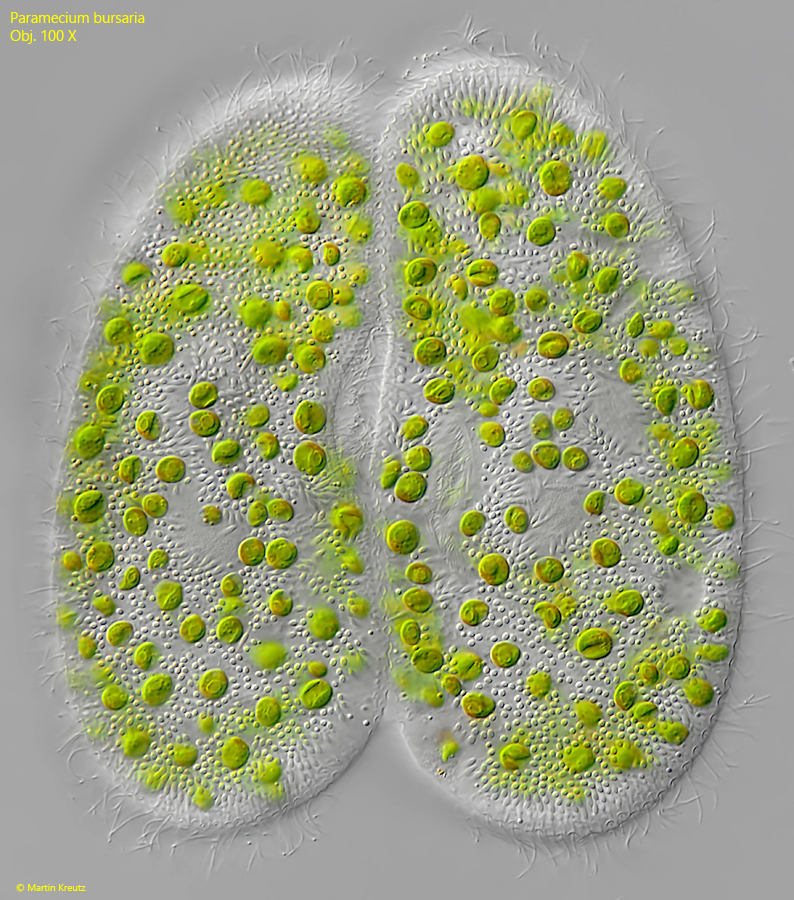
Fig. 11: Paramecium bursaria. L = 129 µm. Two specimens in conjugation. The conjugants are connected via a cytoplasm bridge in the mouth region. Obj. 100 X.
After the cells are connected, the micronuclei in both cells begin to inflate (s. fig. 12).
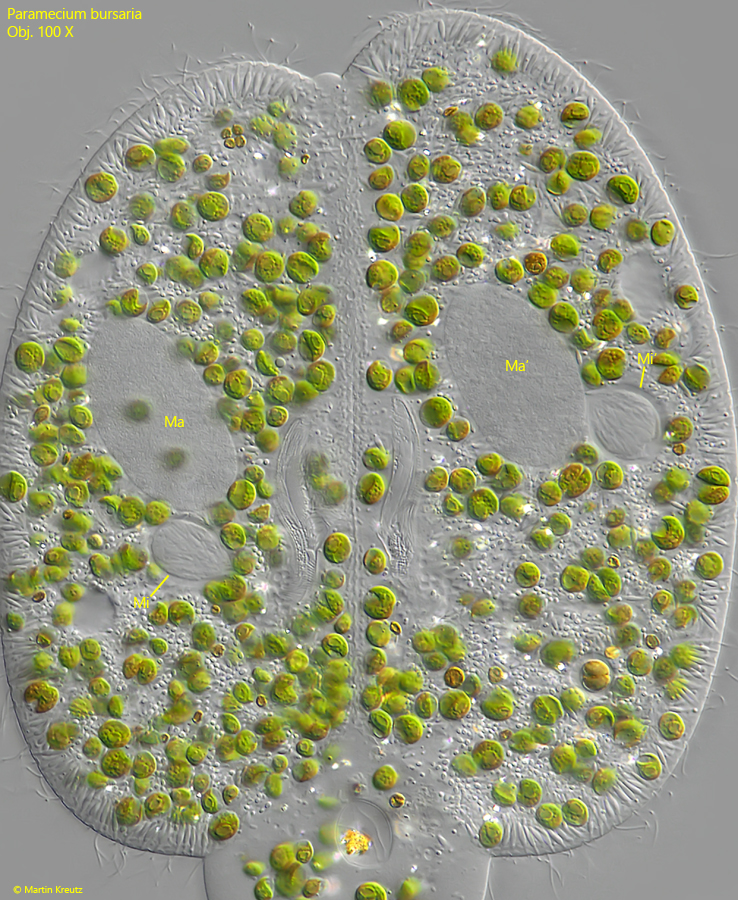
Fig. 12: Paramecium bursaria. After the cells are connected, the micronuclei (Mi, Mi’) in both cells begin to inflate. Ma, Ma’ = macronuclei. Obj. 100 X.
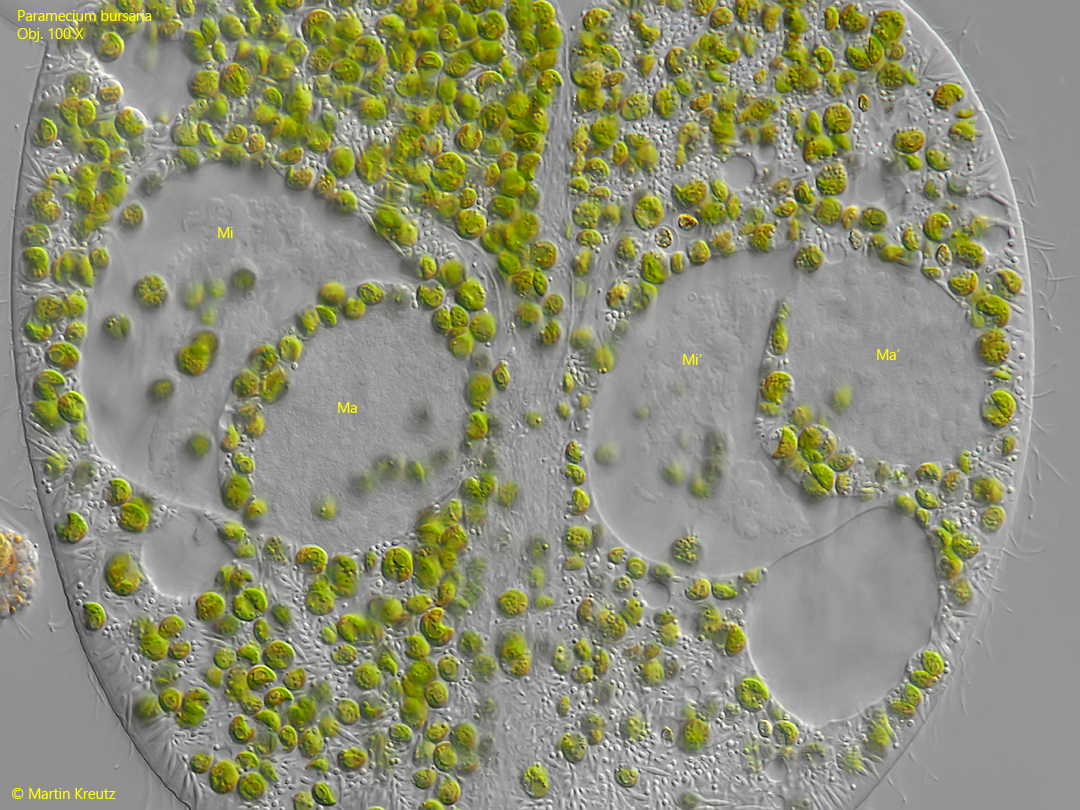
Fig. 13: Paramecium bursaria. Cloud-like structures become visible in the micronuclei (Mi, Mi’) and they take on a crescent shape. This condition corresponds to the drawing of the left cell in fig. 10a. Ma, Ma’ = macronuclei. Obj. 100 X.
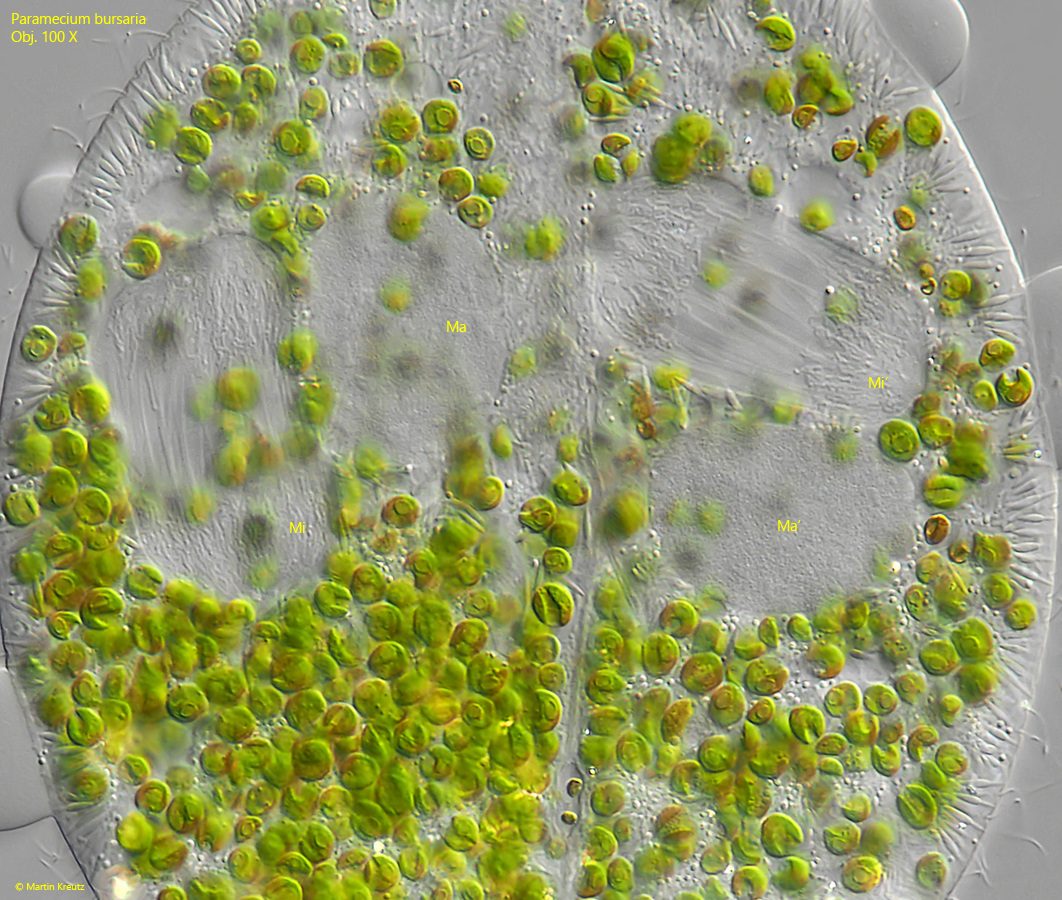
Fig. 14: Paramecium bursaria. The chromosomes organize and the spindle apparatus is formed in the micronuclei (Mi, Mi’). The first meiotic maturation division is initiated. The image corresponds to the drawing of the right cell in fig. 10a. Ma, Ma’ = macronuclei. Obj. 100 X.
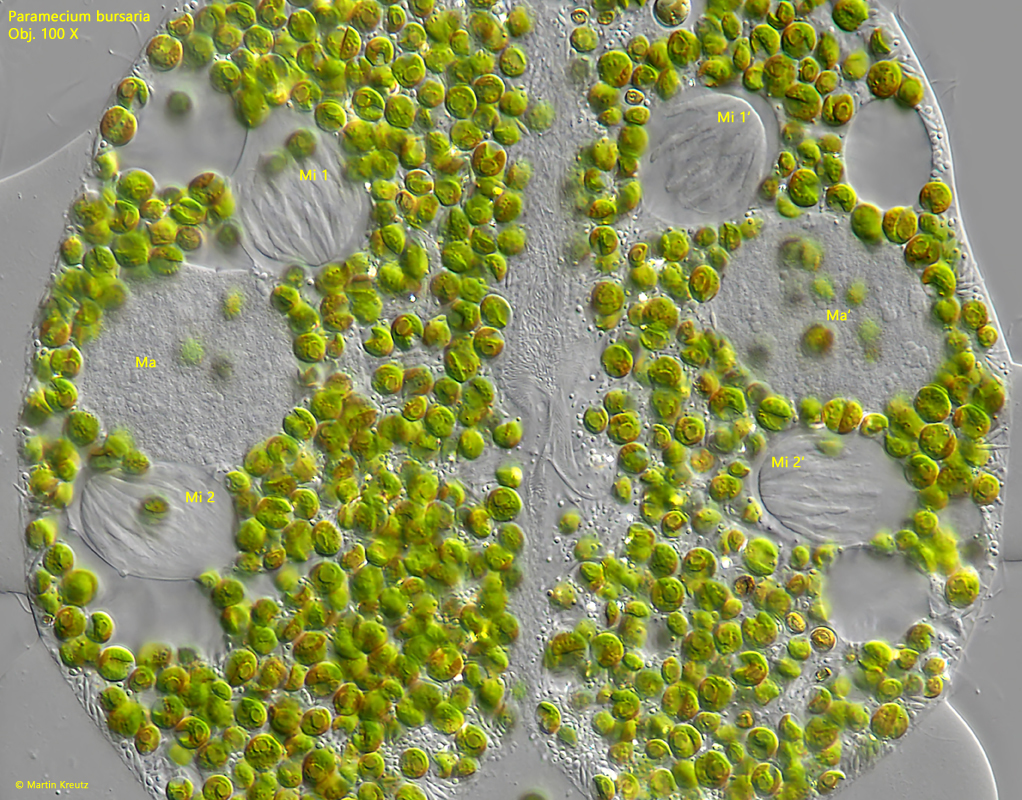
Fig. 15: Paramecium bursaria. The first meiotic maturation division results in two micronuclei in each cell (Mi 1, Mi 2, Mi 1′, Mi 2′), which begin a second meiotic division. This corresponds to drawing 10b. Ma, Ma’ = macronuclei. Obj. 100 X.
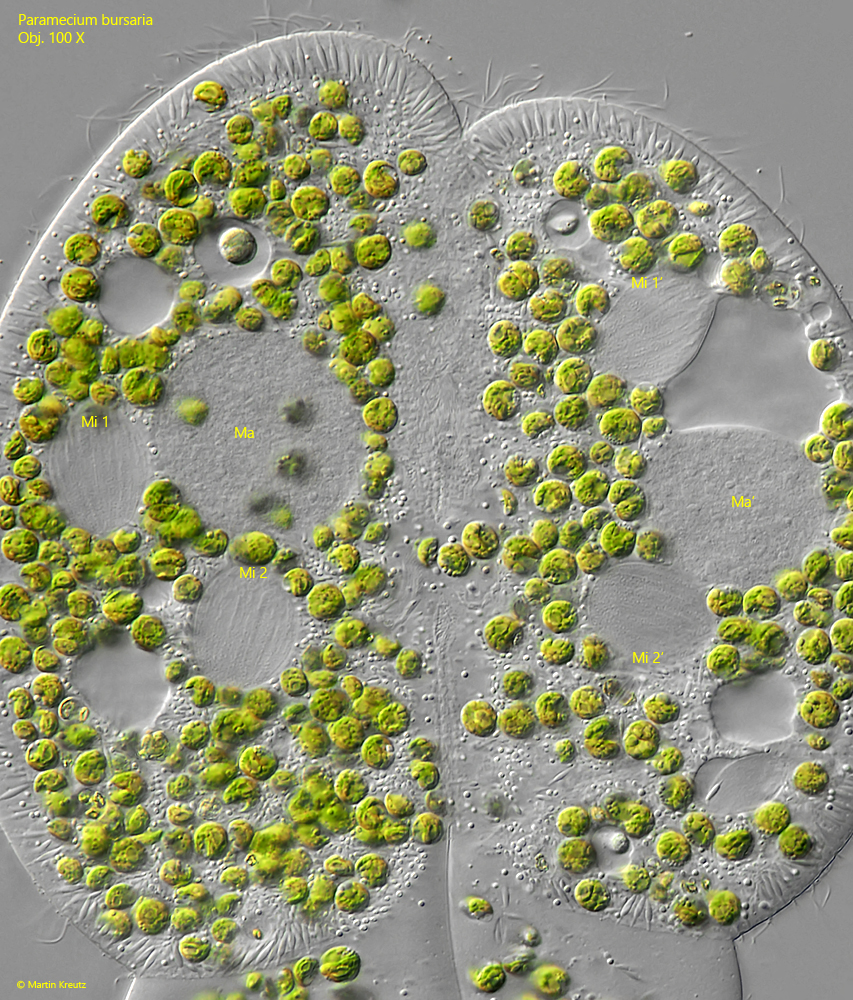
Fig. 16: Paramecium bursaria. This image is showing the same status as shown in fig. 15, but with a visible spindle apparatus (fine striation in the middle of the micronuclei Mi 1, Mi 2, Mi 1′ and Mi 2′). Ma, Ma’ = macronuclei. Obj. 100 X.
After two meiotic divisions four haploid micronuclei are formed per cell. Three of them will be desintegrated. The remaining micronucleus in each cell undergoes another, meiotic division. One of the newly formed micronuclei remains in the parent cell (resting micronucleus), while the second micronucleus is transferred to the partner cell (migrating micronucleus). The transfer of the migrating micronuclei via the cytoplasmic bridge into the partner cell is carried out by strongly extended spindles apparatuses. The extraordinarily long spindle apparatuses can be bend in a U-shape (s. fig. 17).
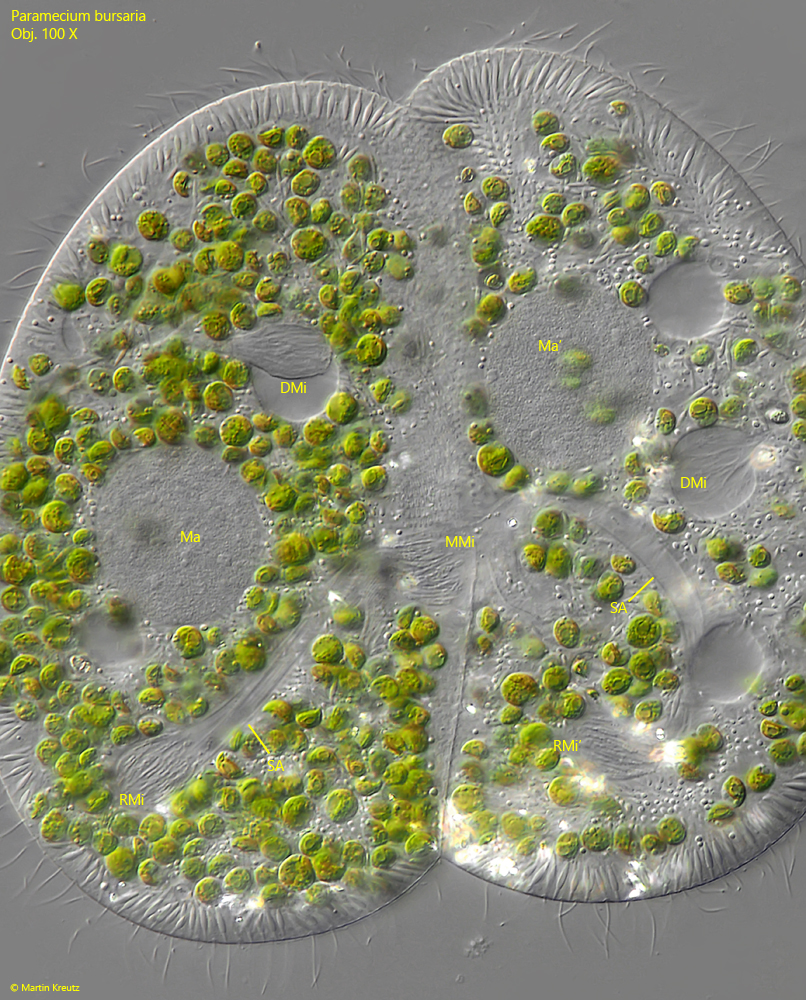
Fig. 17: Paramecium bursaria. After the second meiotic division three micronuclei will be desintegrated. In each cell one of these micronuclei is visible (DMi, DMi’). The remaining micronuclei in each cell undergoes a further meiotic division to form the resting micronucleus (RMi, RMi’) and the migrating micronucleus (MMi). To transfer the migrating micronucleus into the partner cell, the spindle apparatus (SA) is strongly elongated. In the left cell the migrating micronucleus MMi is visible near the cytoplasm bridge between the conjugants. In the right cell the migrating micronuscleus is out of focus but in both cells the elongated spindle apparatuses (SA) are visible. This status of the meiosis corrsponds to the drawing fig. 10c. Ma, Ma’ = macronuclei. Obj. 100 X.
After the exchange of the migrating micronuclei the spindle apparatuses are desintegrated and the exchanged micronuclei fuses with the resting micronuclei to form a new, diploid micronucleus. According my interpretaion the following in fig. 18 shows the two exchanged migratting micronuclei at the cytoplasmic bridge between the conjugants, while the resting micronuclei are located further out in the plasm.
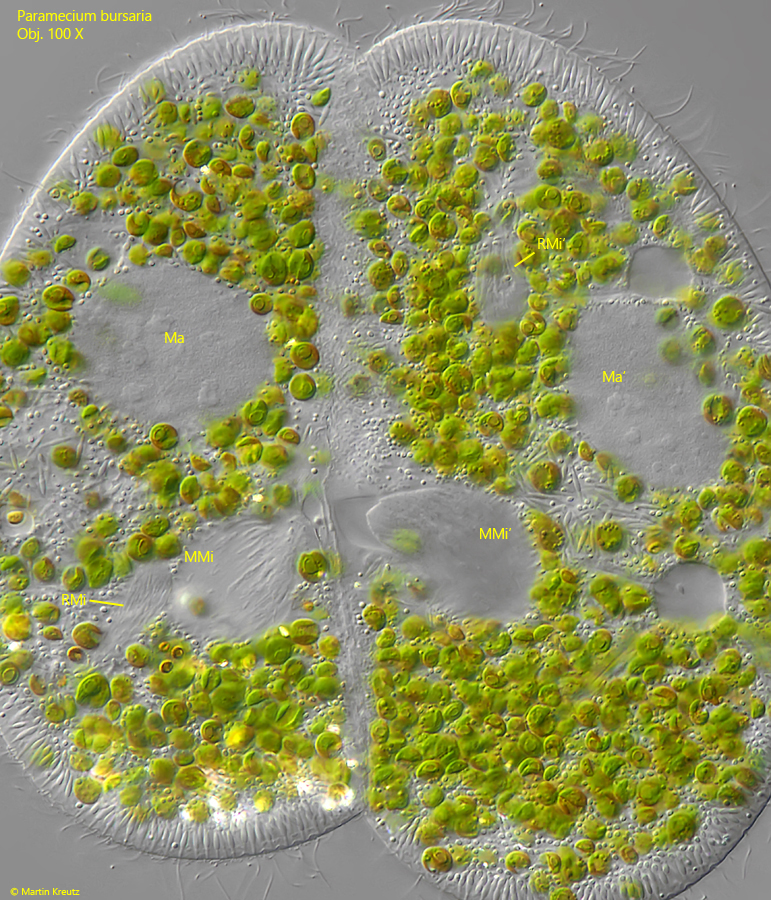
Fig. 18: Paramecium bursaria. The meiosis in the status after the exchange of the migrating micronuclei MMi and MMi’ visible near the cytoplasm bridge between the conjugants. The resting micronuclei RMi and RMi’ are located further out in the plasm. Later the migrating micronuclei will fuse with the resting micronuclei to form a new, diploid micronucleus in each cell. This image corrsponds to drawing fig. 10d. Ma, Ma’ = macronuclei. Obj. 100 X.
In the same sample where I found the high concentration of conjugating pairs, I observed a frequent occurrence of the parasitic bacterium Holospora elegans. This bacterium attacks the micronucleus of host cell and lives in it. The infested specimens can be easily recognized by the spherically inflated micronuclei, which are filled with the spindle-shaped cells of Holospora elegans (s. fig. 19).
Whether the high number of conjugations in the population of Paramecium bursaria is related to the simultaneous infestation with Holospora elegans can only be conjectured. It is conceivable that the infestation with Holospora elegans led to increased sexual activity in order to better counteract the parasite with new genetic variants.
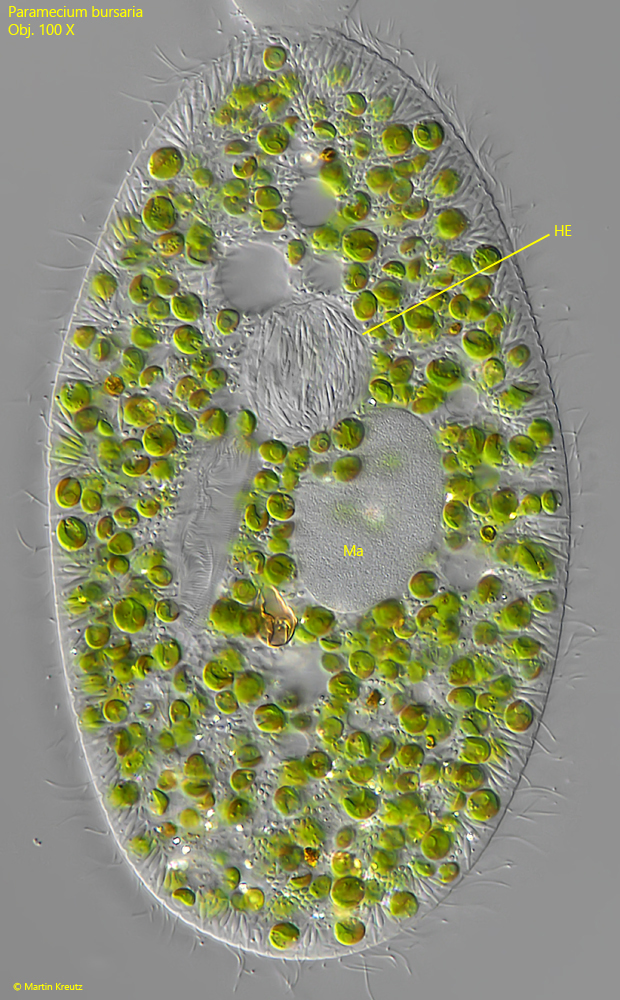
Fig. 19: Paramecium bursaria. A specimen infested with the bacterium Holospora elegans (HE). This bacterium enter the micronucleus and lives in it. Due to the mass of spindle-shaped bacteria the micronucleus is spherically inflated. Ma = macronucleus. Obj. 100 X.