Paramecium caudatum Ehrenberg, 1833
Most likely ID: n.a.
Synonym: n.a.
Sampling location: Simmelried, Ulmisried, Purren pond, Mainau pond, Bündtlisried, Bussenried, Pond of the convent Hegne
Phylogenetic tree: Paramecium caudatum
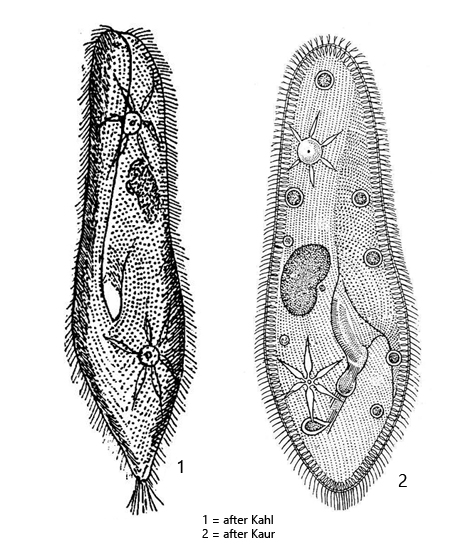
Diagnosis:
- body slipper-shaped, anteriorly rounded, posteriorly conical or narrowly rounded
- length 170–300 µm
- ventral surface with a distinct groove of the oral apparatus, widening anteriorly
- macronucleus ellipsoid in mid-body
- one spherical or broadly oval micronucleus adjacent to macronucleus
- two contractile vacuoles in anterior third and posterior third on dorsal side
- each contractile vacuole with one excretion porus
- contractile vacuoles with about 7 radial collecting ducts each
- spindle-shaped extrusomes (3–4 µm long) forming a distinct seam beneath pellicle
- pellicle quadrangularly fielded
- ciliation uniform, 80–120 ciliary rows of paired cilia
- a tuft of caudal cilia
I find Paramecium caudatum in all of my sampling sites that have a mud layer of decomposed plant material. Paramecium caudatum is found there in the aerobic, upper layer. Paramecium caudatum is practically never found between floating plant masses.
Paramecium caudatum is the best-studied ciliate of all and is kept in culture in thousands of laboratories around the world. In samples from the field, Paramecium caudatum is conspicuous by its size and typical shape, which resembles a slipper. The ciliate is a fast swimmer, but can be easily fixed under the coverslip and then examined in detail. Important features are the two contractile vacuoles with clearly visible collecting ducts and one excretory porus each. A rather large micronucleus with a diameter of about 8 µm is attached to the large macronucleus. The similar species Paramecium aurelia has two small micronuclei.
At the posterior end, Paramecium caudatum has a tuft of caudal cilia, which are about 10–20 µm long. They are characteristically bent outwards. The extrusomes are spindle-shaped and form a dense seam under the pellicle. The cytoplasm is often filled with highly refractive and birefringent crystals.
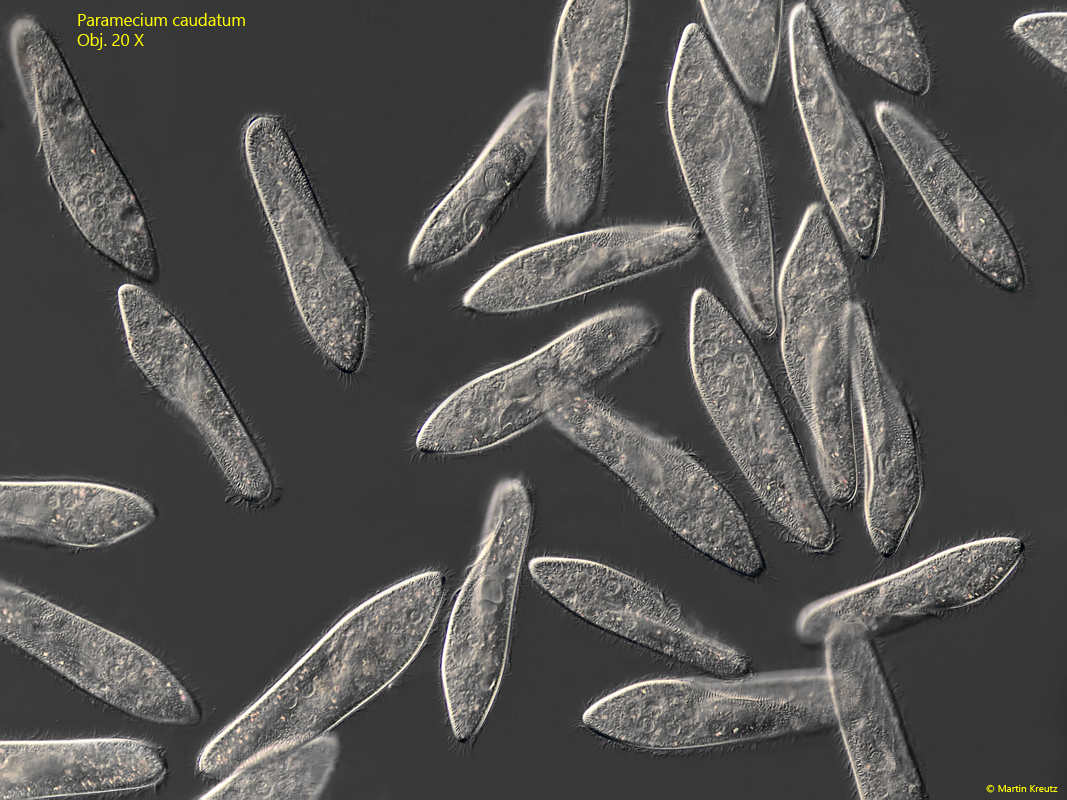
Fig. 1: Paramecium caudatum. L = 200–260 µm. An aggregation of several freely swimming specimens. Obj. 20 X.
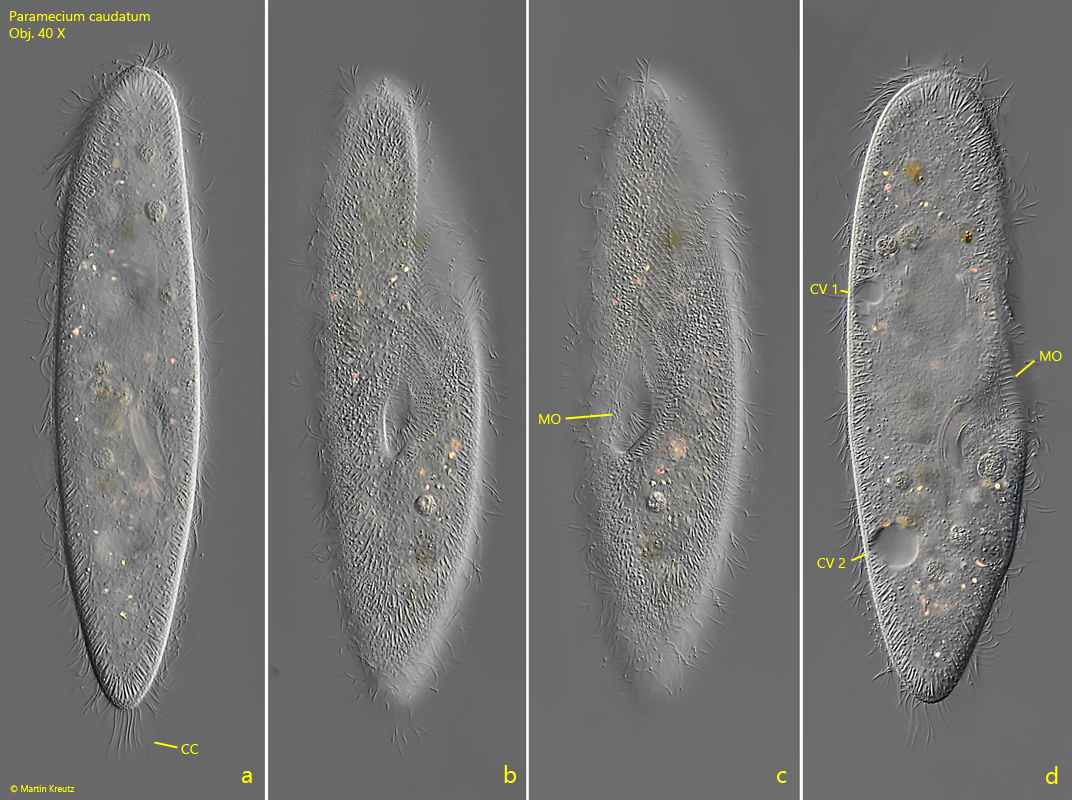
Fig. 2 a-d: Paramecium caudatum. L = 225 µm. A freely swimming specimen from dorsal (a), ventral (b, c) and from right (d). CC = tuft of caudal cilia, CV 1, CV 2 = contractile vacuoles, MO = mouth opening. Obj. 40 X.
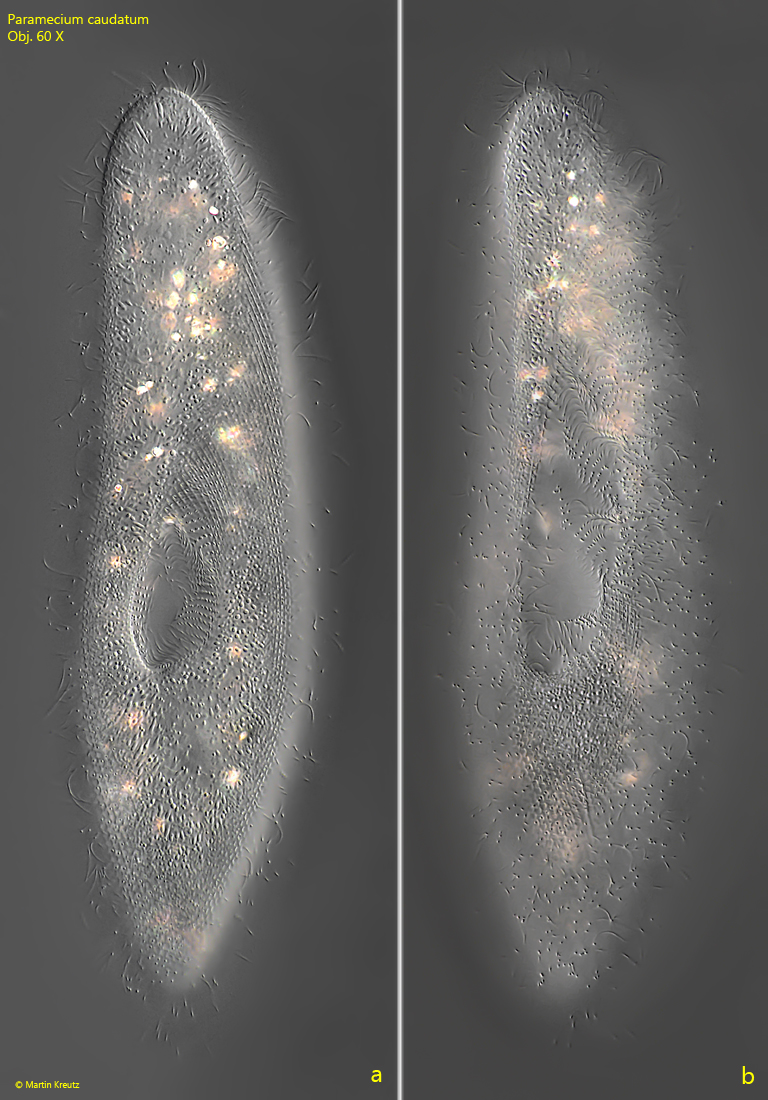
Fig. 3 a-b: Paramecium caudatum. L = 250 µm. A freely swimming specimen from ventral. Note the groove of the oral apparatus widening toward the anterior end. Obj. 60 X.
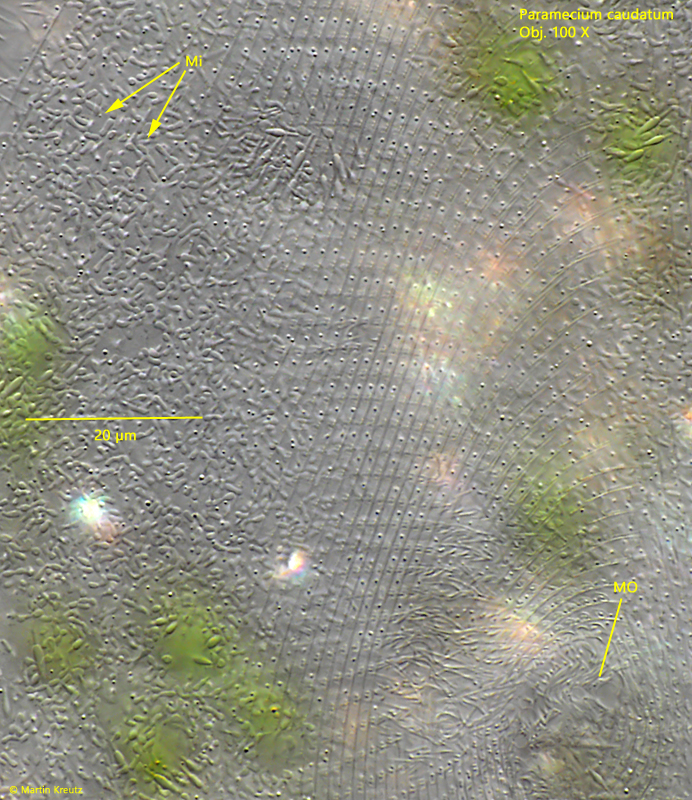
Fig. 4: Paramecium caudatum. The quadrangularly fielded pellicle in front of the mouth opening (MO). Note the numerous mitochondria beneath the pellicle. Obj. 100 X.
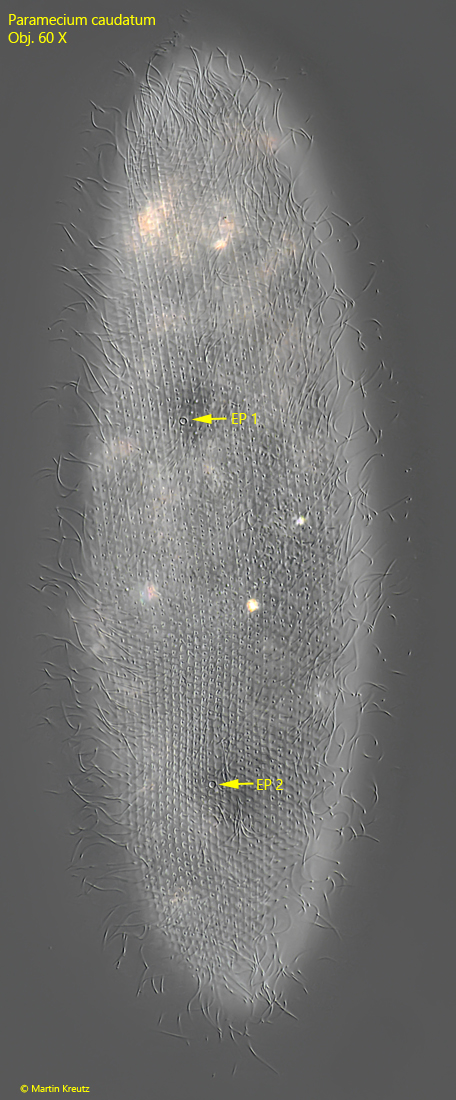
Fig. 5: Paramecium caudatum. L = 242 µm. A slightly squashed specimen from dorsal. Note the two excretory pores of the two contractile vacuoles (EP 1, EP 2). Obj. 100 X.
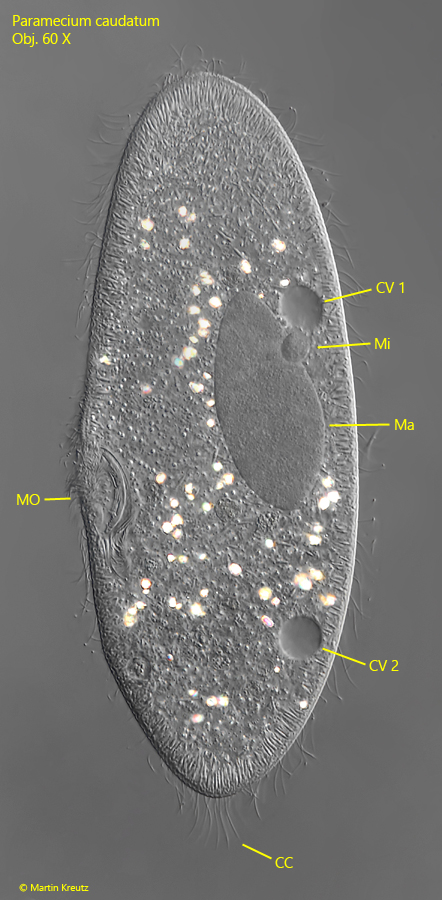
Fig. 6: Paramecium caudatum. The macronucleus (Ma) und the spherical micronucleus (Mi) in a squashed specimen. CC = tuft of caudal cilia, CV 1, CV 2 = contractile vacuoles, MO = mouth opening. Obj. 60 X.
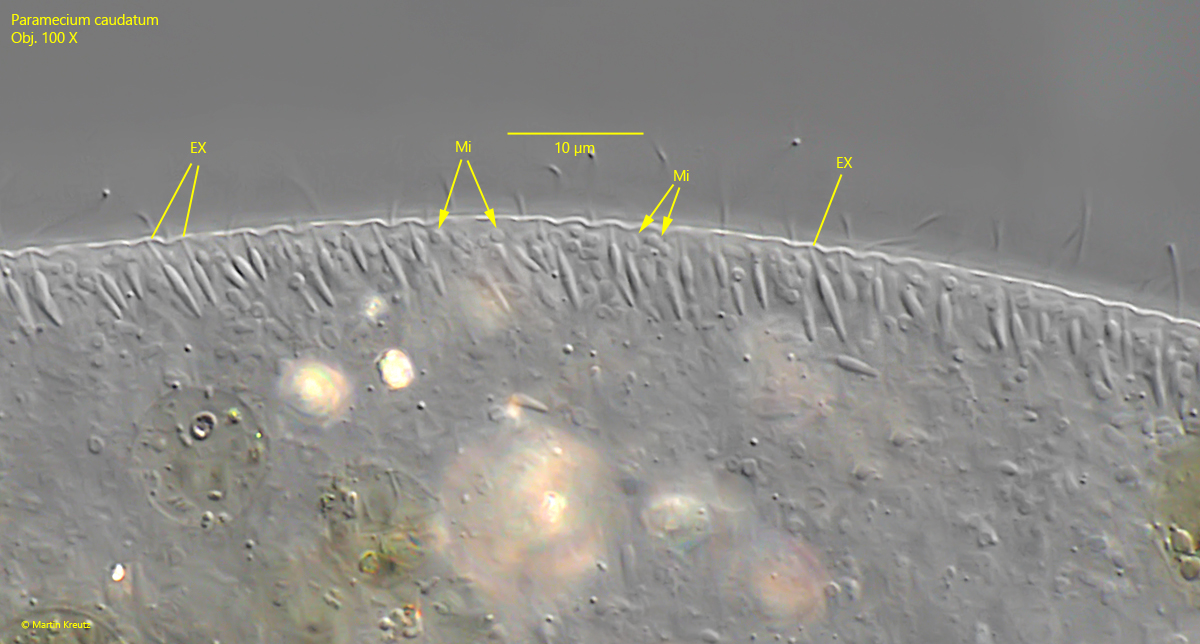
Fig. 7: Paramecium caudatum. The spindle-shaped extrusomes (EX) and the mitochondria (Mi) beneath the pellicle. Obj. 100 X.
In November 2010, I collected old samples taken from the Simmelried in a small 10 liter aquarium instead of throwing them away. To stimulate the growth of bacteria eaters, I added a few drops of condensed milk to the aquarium. After about 1 week, a large population of Paramecium caudatum developed in it. In some specimens, however, I discovered refractive bodies that were obviously filled with densely packed rods (s. fig. 8). At first I thought these bodies were food vacuoles filled with bacteria.
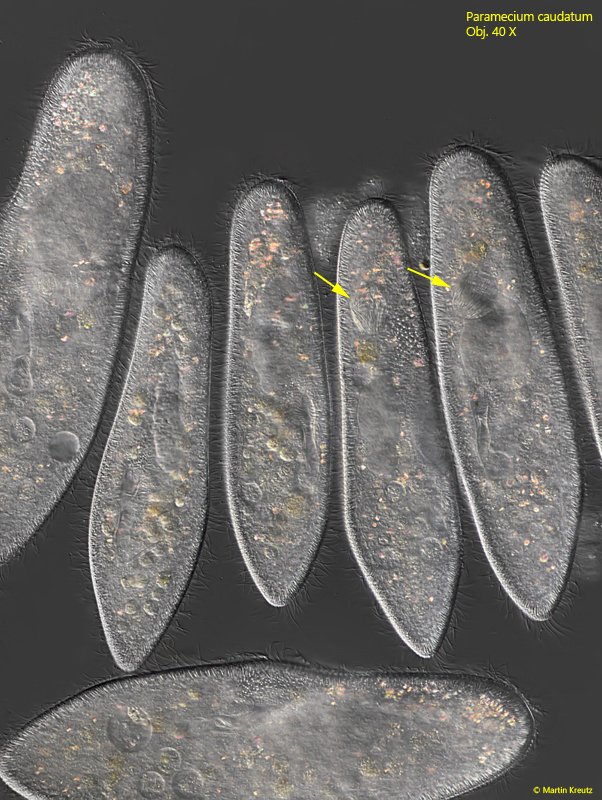
Fig. 8: Paramecium caudatum. Freely swimming specimens of a Simmelried population from November 2010. In some of the specimens highly refractive bodies are visible, densely filled with rods (arrows). Obj. 40 X.
However, I noticed that not all specimens had these highly refractive bodies and when this was the case, these bodies were always found in the same position, namely in the middle of the body or in the front third. I then examined one specimen more closely (s. fig. 9)
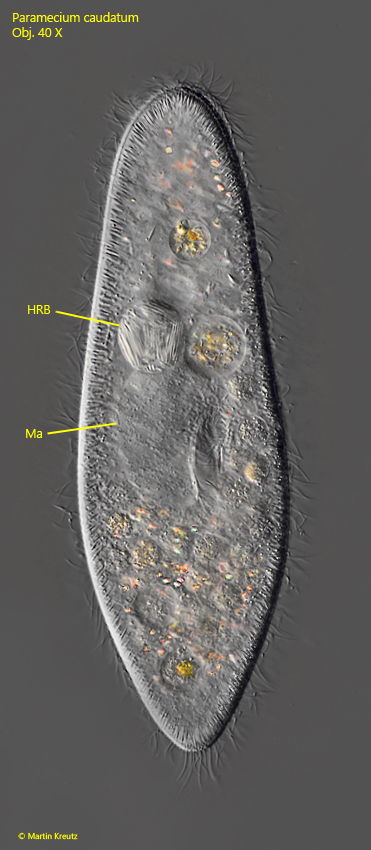
Fig. 9: Paramecium caudatum. L = 227 µm. A specimen with a highly refractive body (HRB), densely filled with rods. The spherical body has a diameter of 25 µm. Ma = macronucleus. Obj. 40 X.
In fact, the supposed food vacuole contained very large, rod-shaped bacteria with a length of 15–20 µm. I examined further specimens and could only ever find one of these bacteria-filled bodies. I also noticed that the micronucleus was missing in all specimens with this highly refractive body and that the macronucleus was often greatly enlarged. These features led me to the correct conclusion that these were infections of the micronucleus with the bacterium Holospora elegans. The identification is based on the shape and size of the bacteria and the fact that only the micronucleus is infected. The similar species Holospora undulata has S-shaped cells.
Holospora elegans has a complex life cycle, which has been studied and described in detail by Goertz & Dieckmann (1980, s. Literture), Goertz & Fujishima (1983, s. Literature) and Fokin & Goertz (2009, s. Literature). Based on the descriptions of these authors, I was able to closely follow and document the life cycle of Holospora elegans in my population of Paramecium caudatum.
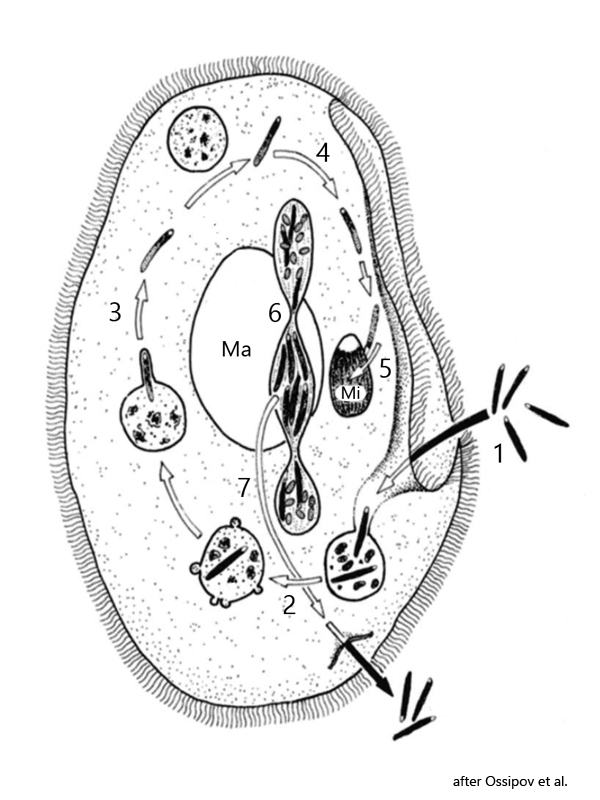
Fig. 10: Schematic life cycle of Holospora elegans.
1 = ingestion of infectious forms of Holospora elegans
2 = transfer of bacteria into food vacuole
3 = release of the infectious form from food vacuole into cytoplasm
4 = active transport of the Holospora cells to the micronucleus
5 = infection of the micronucleus and start of production of invectious and reproductive bacteria cells
6 = during mitosis of micronucleus separation of reproductive bacteria into the both micronuclei and formation of an intermediate body filled with the infectious forms
7 = transfer and release of the intermediate body into the external medium.
Ma = macronucleus, Mi = micronucleus.
The life cycle of Holospora elegans (s. fig. 10) begins with the phagocytosis of infectious cells of Holospora elegans by the host Paramecium caudatum (1). Like all other food particles, the infectious cells first enter a food vacuole (2). Here, Holospora elegans escapes immediate digestion by “reprogramming” the membrane proteins of the food vacuole, which prevents fusion with lysosomes from the surrounding cytoplasm with the food vacuole (3). This reprogramming of the membrane proteins of the food vacuole is host-specific, which means that Holospora cannot escape digestion in the wrong host. In the correct host, however, the transformation of the vacuole membrane into a transport vacuole begins, which wraps itself tightly around the bacterial cell. In this “camouflage suit”, Holospora elegans is actively transported from the host cell to the micronucleus (4). This transport across the actin-myosin scaffold of the cell is induced by Holospora through an 89 kDA protein produced by the bacterial cell. Once at the micronucleus, the bacterium penetrates the nuclear membrane and immediately begins cell division, forming smaller, reproductive and larger, infectious cells (5). This transformation of the external cell form depending on the stage of infection is called dimorphism.
The smaller, reproductive cells are oval and only approx. 2 µm long, while the infectious cells are 10-20 µm long rods with pointed ends. During the growth phase, the micronucleus of Paramecium caudatum can swell to ten times its original size. The genetic material of Paramecium caudatum is obviously not damaged. In order to leave the host cell and enter other host cells, Holospora elegans has developed a “dual” mechanism that ensures that when a Paramecium caudatum cell divides, the daughter cell is also infected with Holospora elegans and that the infectious forms formed in the micronucleus are released to be phagocytosed by other host cells. This involves overcoming the host’s nuclear and cell membranes.
This feat takes place when Paramecium caudatum begins to divide. A mitotic spindle then forms within the micronucleus. However, not only the chromosomes are organized and separated, but also the cells of Holospora elegans. The reproductive forms are drawn very selectively to the poles of the mitotic spindle, while the large, infectious forms are collected in the equatorial plane of the micronucleus in a so-called intermediate body (6). The two newly formed micronuclei, which are mainly filled with reproductive, small cells, are distributed to the daughter cells (which are then both infected), while the intermediate body with the infectious forms is expelled through the pellicle (7). With this ingenious mechanism, Holospora elegans manages to enter both daughter cells as well as to release new, infectious forms into the environment to enable new infections.
In my infected population of Paramecium caudatum, I was able to capture the moment after the infectious cell of Holospora elegans has just entered the micronucleus and before the cell divisions has started (s. fig. 11). This phase of the life cycle lasts a maximum of 24 hours and many infected host cells have to be examined to find this stage.

Fig. 11: Paramecium caudatum. The micronucleus (Mi) is infected with the first cell of Holospora elegans (HE). The cell division of the bacterium in the micronucleus has not started in this phase. Ma = macronucleus. Obj. 100 X.
The invaded, infectious cell of Holospora elegans begins cell division in the micronucleus. The newly formed bacterial cells continue to fill the micronucleus and later enlarge it. The division activities of Holospora elegans require a higher metabolism of the host cell, which also increases the size of the macronucleus. This often results in flocculent aggregates in the macronucleus (s. fig. 12).
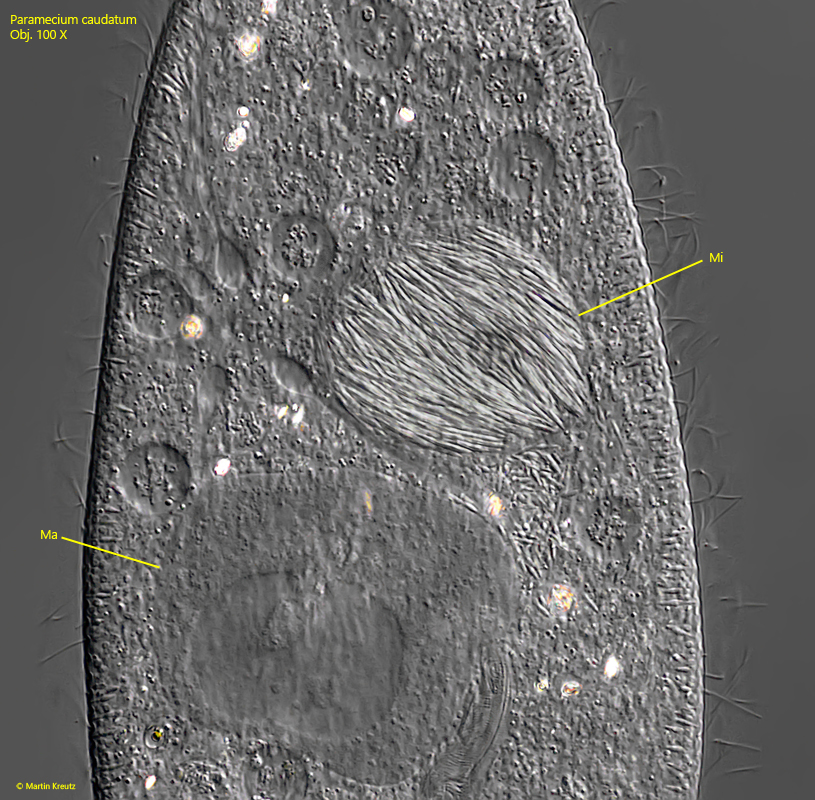
Fig. 12: Paramecium caudatum. A squashed specimen whose micronucleus (Mi) is densely filled with rod-shaped cells of the bacterium Holospora elegans after numerous cell divisions of the bacterium. Note the enlarged macronucleus (Ma). Obj. 100 X.
The next phase of the life cycle of Holospora elegans begins with the cell division of the host cell. Prior to this, not only the rod-shaped, infectious cells of Holospora elegans have formed in the micronucleus, but also small, oval cells, which are referred to as reproductive cells, as they are distributed to the daughter cells during cell division. If infected cells of Paramecium caudatum are squashed strongly, the reproductive and infectious cells of Holospora elegans are released (s. fig. 13).

Fig. 13: Holospora elegans.The infectious (IC) and reproductive cells (RC) released from a strongly squashed specimen of Paramecium caudatum. Obj. 100 X.
At the beginning of the mitotic division of the micronucleus, the centrosomes are created at opposite poles, from which the spindle apparatus is formed. The chromosomes are then pulled towards the two poles. In the micronucleus infected with Holospora elegans, however, the reproductive bacterial cells are also drawn to the poles, while a concentration of the large, infectious bacterial cells remains in the center (s. fig. 14).
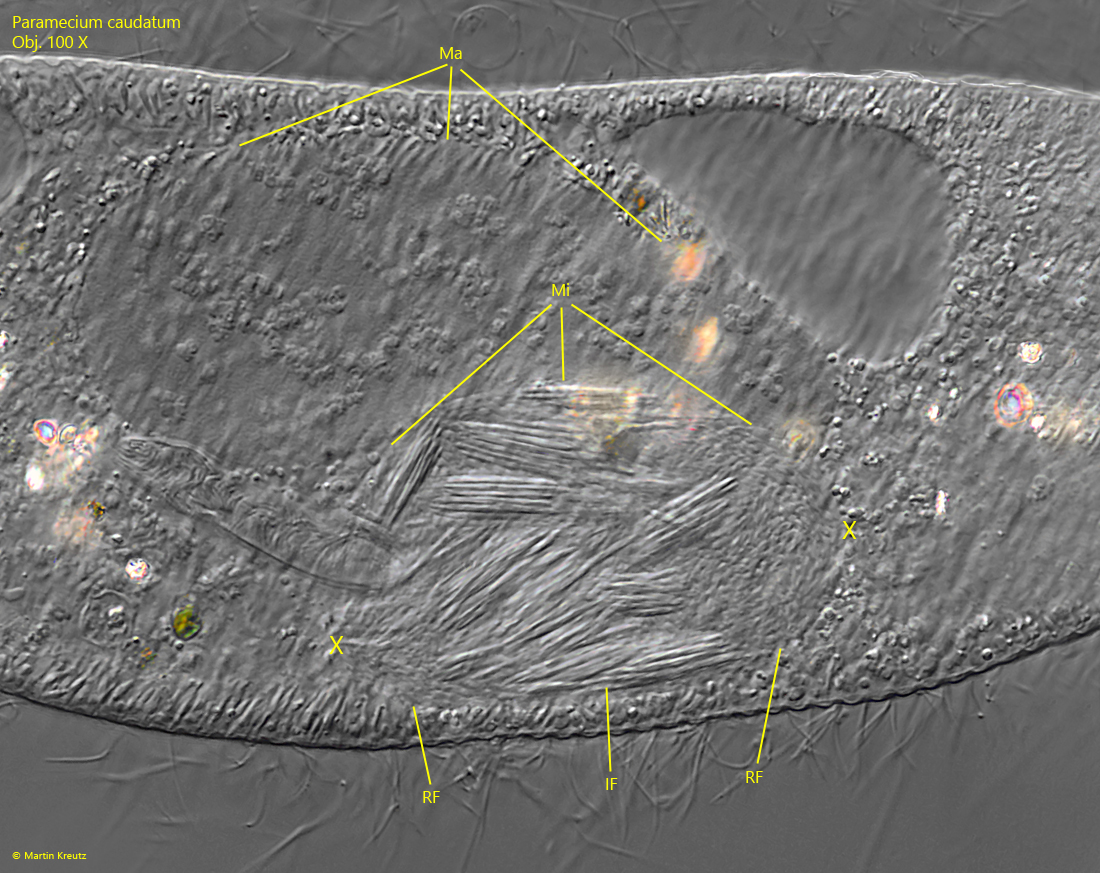
Fig. 14: Paramecium caudatum.The reproductive forms (RF) of Holospora elegans are drawn to the poles of the spindle apparatus (marked with X) and concentrate there, while the larger, infectious forms (IF) remain in the center. Ma = macronucleus, Mi = micronucleus. Obj. 100 X.
When the spindle apparatus separates the two new micronuclei, the reproductive forms of Holospora elegans that previously accumulated at the poles are pulled along and later enter the two daughter cells in this way. At the same time, the infectious forms remain in the middle of the spindle apparatus, where they form the so-called intermediate body (IB, s. figs. 15 and 16). After complete separation of the new micronuclei and degradation of the spindle apparatus, the intermediate body is excreted from the cell and enters the surrounding medium, where the infectious cells can be phagocytized by other host cells.

Fig. 15: Paramecium caudatum. In this phase of mitosis, the spindle apparatus has already largely separated the two new micronuclei (Mi 1, Mi 2) and the intermediate body (IB) with the infectious forms remains in the middle. After degradation of the spindle apparatus, the intermdiate body is excreted from the cell together with the infectious forms it contains before the actual cell division begins. Ma 1, Ma 2 = macronuclei. Obj. 100 X.

Fig. 16: Paramecium caudatum. The separation of infectious and reproductive cells is not completely selective, as some long rods of the infectious form of Holospora elegans can also be recognized in the newly formed micronucleus (Mi). The fibers of the spindle apparatus (FSA) can be seen between the intermediate body IB) and the new micronucleus. Obj. 100 X.
During mitosis of the micronucleus of Paramecium caudatum, the presence of Holospora elegans often causes division abnormalities. The spindle apparatus then does not elongate sufficiently so that both daughter micronuclei remain in one cell. Or abnormal spindle apparatuses are created with 4 instead of 2 poles. Sometimes a part of the intermediate body with the infectious forms remains in the host cell. This manifests itself in findings of Paramecium caudatum cells that contain 4–8 or even more infected micronuclei with Holospora elegans. During my investigation of the infected population I found several of these abnormal cells (s. figs. 17 and 18).
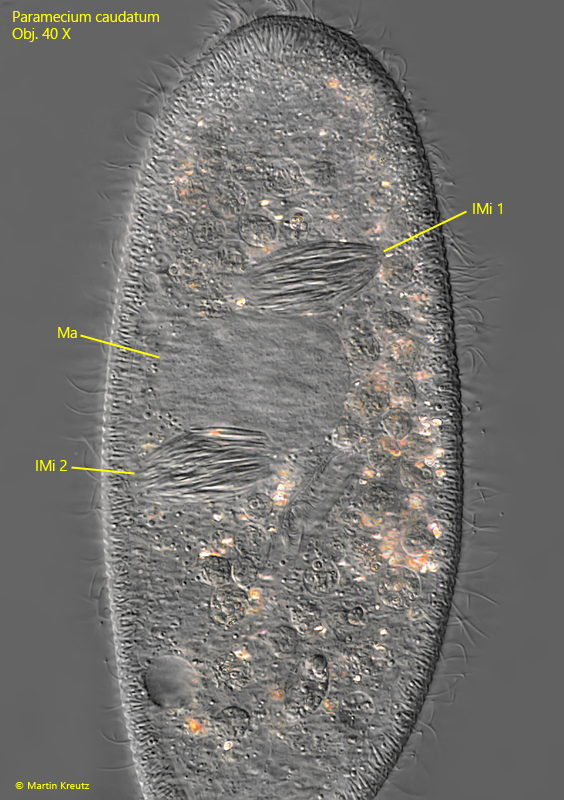
Fig. 17: Paramecium caudatum. A specimen with two micronuclei (IMi 1, IMi 2) infected with Holospora elegans. Ma = macronucleus. Obj. 40 X.
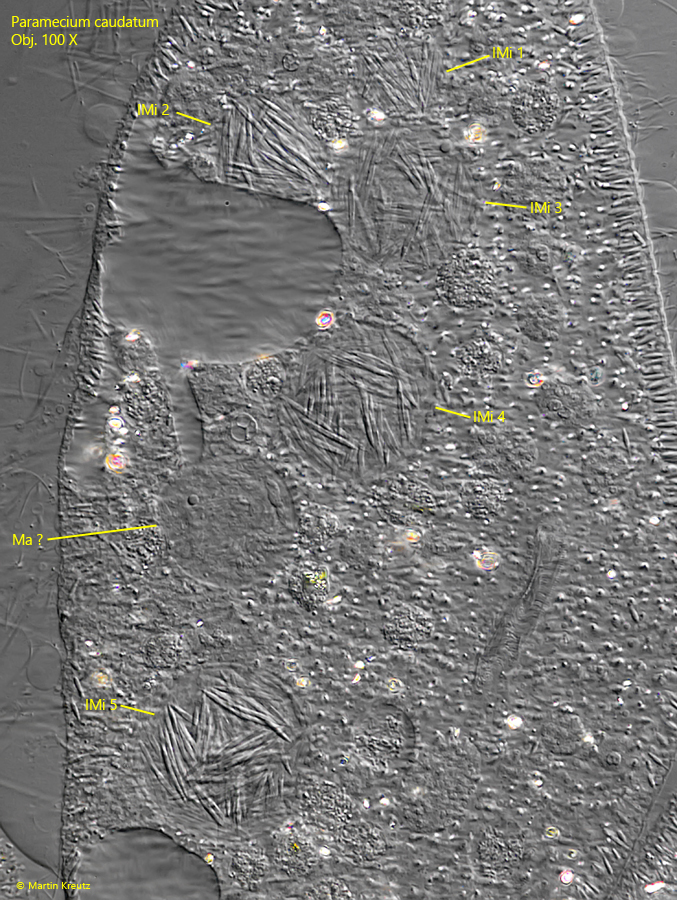
Fig. 18: Paramecium caudatum. A specimen with five micronuclei (IMi 1–5) infected with Holospora elegans. Ma ? = probably the macronucleus. Obj. 100 X.
The close relationship between Paramecium caudatum and Holospora elegans testifies to a perfect evolutionary adaptation that must have taken place over a very long period of time. After all, Holospora elegans lives and reproduces in the micronucleus, the reproductive core of the host, without obviously damaging it. This relationship has been studied intensively and no direct detrimental effects on Paramecium caudatum have been found. Antibiotics can completely eliminate Holospora elegans from an infected Paramecium population without any effect on the host. This means that the entire life cycle of Holospora elegans is closely linked to that of Paramecium caudatum, but not vice versa.
So far, it has not been possible to cultivate Holospora elegans outside of Paramecium caudatum, so it is not entirely clear what type of releationship is present here. However, there is an indication that this is not a symbiosis. Laboratory experiments have shown that Paramecium caudatum has defense mechanisms to prevent infection with Holospora elegans and that complete infection of a Paramecium caudatum population with Holospora elegans cannot be achieved because the bacterial cells in the host’s cytoplasm can be eliminated (Fokin, Görtz, 2009). One reason for this defense reaction could be that infected specimens of Paramecium caudatum are no longer capable of conjugation, which represents an effective selection disadvantage.
In the same population of Paramecium caudatum in which I was able to observe the infections with Holospora elegans, I also found many specimens with unusually large crystals in the cytoplasm (s. fig. 19 a-b). These specimens also all had a strongly deformed macronucleus with crater-shaped depressions. It was striking that these large crystals were never found in specimens infested with Holospora elegans.
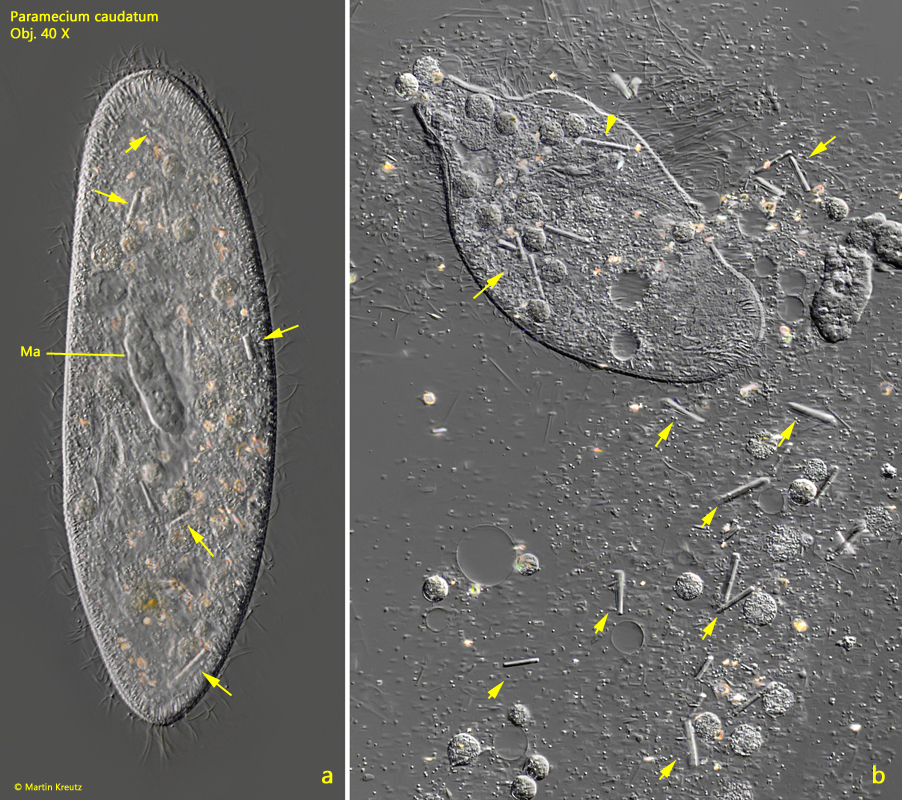
Fig. 19 a-b: Paramecium caudatum. A slightly (a) and a strongly squashed specimen (b) with unusual large crystals scattered in the cytoplasm (arrows). Note the deformed macronucleus (Ma). Obj. 40 X.
One could assume that this is ingested food or the storage crystals often found in Paramecium caudatum. It can be ruled out that the crystals were ingested as food, as they are not found in food vacuoles. The common storage crystals of Paramecium caudatum are mostly drusen-shaped and birefringent, which is why they appear colored in the DIC. However, the long, rod-shaped crystals were not only unusually large at 15–25 µm in length, but also not birefringent (s. figs. 20 and 21). In addition, they were only present in individual specimens, which always showed the deformed macronucleus.The micronucleus was also present in these specimens but slightly enlarged or deformed.
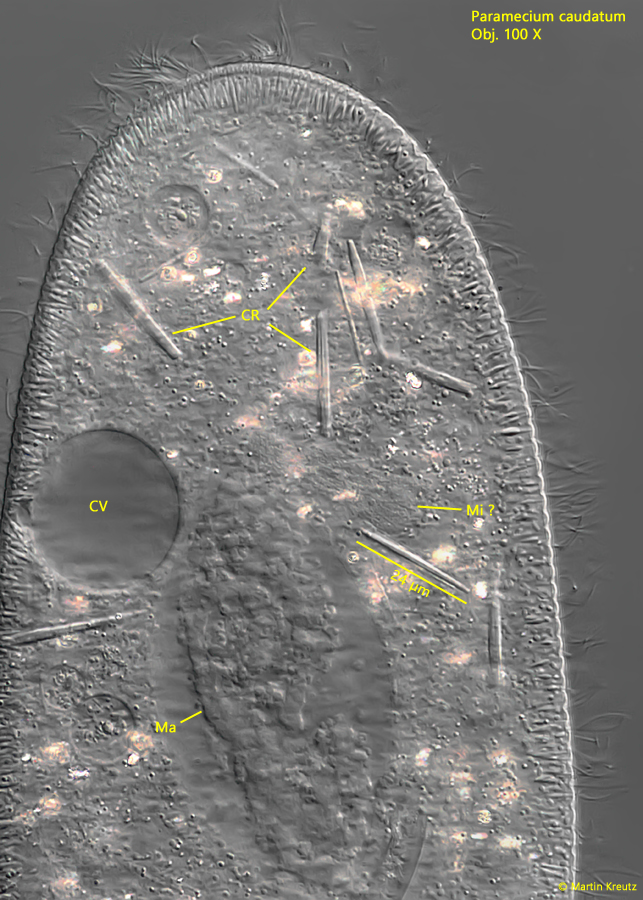
Fig. 20: Paramecium caudatum. A squashed specimen with several of the large, rod-shaped crystals (CR). Note the deformed macronucleus (Ma). CV = contractile vacuole, Mi ?= probably the micronucleus. Obj. 100 X.

Fig. 21: Paramecium caudatum. A second squashed specimen with several of the large, rod-shaped crystals (CR). Ma = macronucleus, Mi = micronucleus. Obj. 100 X.
In a strongly squashed specimens, I was able to observe that the crystals break under the pressure of the coverslip. However, this results in unusual fracture lines, which usually run through the crystal in an S-shape (s. fig. 22). I could not find descriptions of comparable crystals found in Paramecium caudatum in the literature. It remains unclear whether this is the crystallization of an unusual metabolic product or perhaps even virus crystals. I never found these kind of crystals again since 2010.
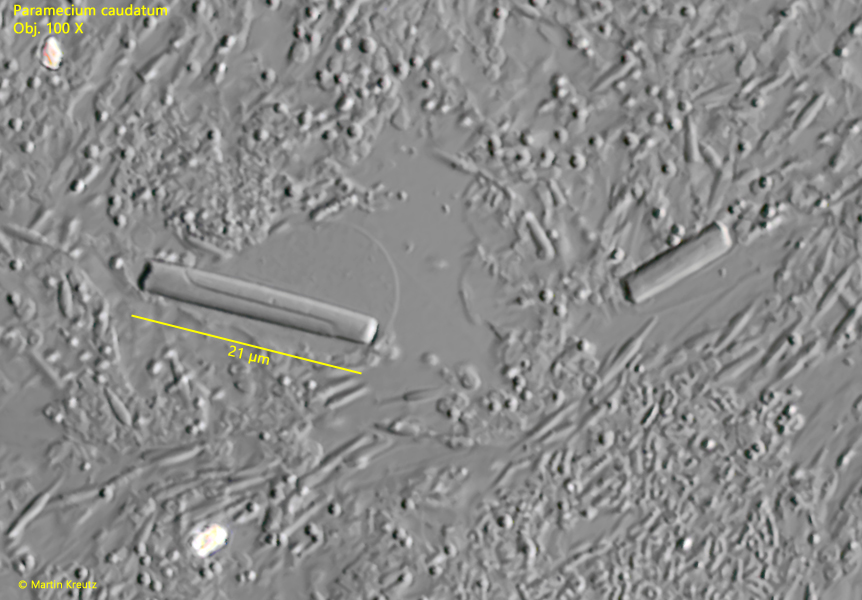
Fig. 22: Paramecium caudatum. Under strong pressure of the coverslip the rod-shaped crystals start to crack and S-shaped fracture lines become visible (s. left crystal). Obj. 100 X.